Minimal brain PBPK model to support the preclinical and clinical development of antibody therapeutics for CNS diseases
- PMID: 34378151
- PMCID: PMC8604880
- DOI: 10.1007/s10928-021-09776-7
Minimal brain PBPK model to support the preclinical and clinical development of antibody therapeutics for CNS diseases
Abstract
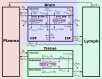
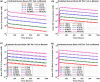
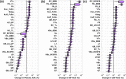
There are several antibody therapeutics in preclinical and clinical development, industry-wide, for the treatment of central nervous system (CNS) disorders. Due to the limited permeability of antibodies across brain barriers, the quantitative understanding of antibody exposure in the CNS is important for the design of antibody drug characteristics and determining appropriate dosing regimens. We have developed a minimal physiologically-based pharmacokinetic (mPBPK) model of the brain for antibody therapeutics, which was reduced from an existing multi-species platform brain PBPK model. All non-brain compartments were combined into a single tissue compartment and cerebral spinal fluid (CSF) compartments were combined into a single CSF compartment. The mPBPK model contains 16 differential equations, compared to 100 in the original PBPK model, and improved simulation speed approximately 11-fold. Area under the curve ratios for minimal versus full PBPK models were close to 1 across species for both brain and plasma compartments, which indicates the reduced model simulations are similar to those of the original model. The minimal model retained detailed physiological processes of the brain while not significantly affecting model predictability, which supports the law of parsimony in the context of balancing model complexity with added predictive power. The minimal model has a variety of applications for supporting the preclinical development of antibody therapeutics and can be expanded to include target information for evaluating target engagement to inform clinical dose selection.
Keywords: Antibody; Drug development; Neuroscience; PBPK; Pharmacokinetics.
© 2021. The Author(s).
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

