Application of Stem Cell Therapy for ACL Graft Regeneration
- PMID: 34381504
- PMCID: PMC8352687
- DOI: 10.1155/2021/6641818
Application of Stem Cell Therapy for ACL Graft Regeneration
Abstract
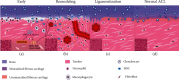
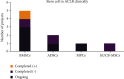
Graft regeneration after anterior cruciate ligament (ACL) reconstruction surgery is a complex three-stage process, which usually takes a long duration and often results in fibrous scar tissue formation that exerts a detrimental impact on the patients' prognosis. Hence, as a regeneration technique, stem cell transplantation has attracted increasing attention. Several different stem cell types have been utilized in animal experiments, and almost all of these have shown good capacity in improving tendon-bone regeneration. Various differentiation inducers have been widely applied together with stem cells to enhance specific lineage differentiation, such as recombinant gene transfection, growth factors, and biomaterials. Among the various different types of stem cells, bone marrow-derived mesenchymal stem cells (BMSCs) have been investigated the most, while ligament stem progenitor cells (LDSCs) have demonstrated the best potential in generating tendon/ligament lineage cells. In the clinic, 4 relevant completed trials have been reported, but only one trial with BMSCs showed improved outcomes, while 5 relevant trials are still in progress. This review describes the process of ACL graft regeneration after implantation and summarizes the current application of stem cells from bench to bedside, as well as discusses future perspectives in this field.
Copyright © 2021 Canlong Wang et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
Similar articles
-
Stem cell therapy: a promising biological strategy for tendon-bone healing after anterior cruciate ligament reconstruction.Cell Prolif. 2016 Apr;49(2):154-62. doi: 10.1111/cpr.12242. Epub 2016 Mar 1. Cell Prolif. 2016. PMID: 26929145 Free PMC article. Review.
-
Exogenous stromal derived factor-1 releasing silk scaffold combined with intra-articular injection of progenitor cells promotes bone-ligament-bone regeneration.Acta Biomater. 2018 Apr 15;71:168-183. doi: 10.1016/j.actbio.2018.02.019. Epub 2018 Mar 7. Acta Biomater. 2018. PMID: 29524675
-
Local administration of TGFβ-1/VEGF165 gene-transduced bone mesenchymal stem cells for Achilles allograft replacement of the anterior cruciate ligament in rabbits.Biochem Biophys Res Commun. 2011 Mar 11;406(2):204-10. doi: 10.1016/j.bbrc.2011.02.015. Epub 2011 Feb 15. Biochem Biophys Res Commun. 2011. PMID: 21303664
-
Functional regeneration of ligament-bone interface using a triphasic silk-based graft.Biomaterials. 2016 Nov;106:180-92. doi: 10.1016/j.biomaterials.2016.08.012. Epub 2016 Aug 9. Biomaterials. 2016. PMID: 27566867
-
Current Evidence of Adult Stem Cells to Enhance Anterior Cruciate Ligament Treatment: A Systematic Review of Animal Trials.Arthroscopy. 2018 Jan;34(1):331-340.e2. doi: 10.1016/j.arthro.2017.07.010. Epub 2017 Sep 28. Arthroscopy. 2018. PMID: 28967542
Cited by
-
Integrating Modern Technologies into Traditional Anterior Cruciate Ligament Tissue Engineering.Bioengineering (Basel). 2025 Jan 7;12(1):39. doi: 10.3390/bioengineering12010039. Bioengineering (Basel). 2025. PMID: 39851313 Free PMC article. Review.
-
Comparative effect of skeletal stem cells versus bone marrow mesenchymal stem cells on rotator cuff tendon-bone healing.J Orthop Translat. 2024 Jun 20;47:87-96. doi: 10.1016/j.jot.2024.05.005. eCollection 2024 Jul. J Orthop Translat. 2024. PMID: 39007033 Free PMC article.
-
Advances in Regenerative Sports Medicine Research.Front Bioeng Biotechnol. 2022 May 13;10:908751. doi: 10.3389/fbioe.2022.908751. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35646865 Free PMC article. Review.
-
ACL injury management: a comprehensive review of novel biotherapeutics.Front Bioeng Biotechnol. 2024 Nov 22;12:1455225. doi: 10.3389/fbioe.2024.1455225. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39650235 Free PMC article. Review.
References
-
- Risberg M. A., Lewek M., Snyder-Mackler L. A systematic review of evidence for anterior cruciate ligament rehabilitation: how much and what type? Physical Therapy in Sport. 2004;5(3):125–145. doi: 10.1016/j.ptsp.2004.02.003. - DOI
-
- Florida S. E., VanDusen K. W., Mahalingam V. D., et al. In vivo structural and cellular remodeling of engineered bone-ligament-bone constructs used for anterior cruciate ligament reconstruction in sheep. Connective Tissue Research. 2016;57(6):526–538. doi: 10.1080/03008207.2016.1187141. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Medical

