Upper respiratory tract SARS-CoV-2 RNA loads in symptomatic and asymptomatic children and adults
- PMID: 34384874
- PMCID: PMC8349738
- DOI: 10.1016/j.cmi.2021.08.001
Upper respiratory tract SARS-CoV-2 RNA loads in symptomatic and asymptomatic children and adults
Abstract
Objectives: Studies comparing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA load in the upper respiratory tract (URT) between children and adults-who either presented with coronavirus disease 2019 (COVID-19) or were asymptomatic-have yielded inconsistent results. Here, we conducted a retrospective, single-centre study to address this issue.
Patients and methods: Included were 1184 consecutive subjects (256 children and 928 adults) testing positive for SARS-CoV-2 RNA in nasopharyngeal exudates (NPs); of these, 424 (121 children and 303 adults) had COVID-19 and 760 (135 children and 625 adults) were asymptomatic close contacts of COVID-19 patients. SARS-CoV-2 RNA testing was carried out using the TaqPath COVID-19 Combo Kit (Thermo Fisher Scientific, MS, USA). The AMPLIRUN® TOTAL SARS-CoV-2 RNA Control (Vircell SA, Granada, Spain) was used for estimating SARS-CoV-2 RNA loads (in copies/mL). SARS-CoV-2 RNA loads at the time of laboratory diagnosis (single specimen/patient) were used for comparison purposes.
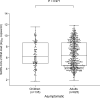
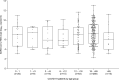
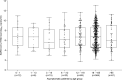
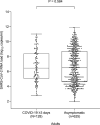
Results: Median initial SARS-CoV-2 RNA load was lower (p 0.094) in children (6.98 log10 copies/mL, range 3.0-11.7) than in adults (7.14 log10 copies/mL, range 2.2-13.4) with COVID-19. As for asymptomatic individuals, median SARS-CoV-2 RNA load was comparable (p 0.97) in children (6.20 log10 copies/mL, range 1.8-11.6) and adults (6.48 log10 copies/mL, range 1.9-11.8). Children with COVID-19 symptoms displayed SARS-CoV-2 RNA loads (6.98 log10 copies/mL, range 3.0-11.7) comparable to those of their asymptomatic counterparts (6.20 log10 copies/mL, range 1.8-11.6) (p 0.61). Meanwhile in adults, median SARS-CoV-2 RNA load was significantly higher in symptomatic (7.14 log10 copies/mL, range 2.2-13.4) than in asymptomatic subjects (6.48 log10 copies/mL, range 1.9-11.8) (p < 0.001). Overall, the observed URT SARS-CoV-2 RNA clearance rate was faster in children than in adults.
Conclusions: Based on viral load data at the time of diagnosis, our results suggest that SARS-CoV-2-infected children, with or without COVID-19, may display NP viral loads of comparable magnitude to those found in their adult counterparts. However, children may have shorter viral shedding than adults.
Keywords: Adults; COVID-19; Children; SARS-CoV-2 RNA; Upper respiratory tract; Viral load.
Copyright © 2021 European Society of Clinical Microbiology and Infectious Diseases. Published by Elsevier Ltd. All rights reserved.
Figures




References
-
- Wölfel R., Corman V.M., Guggemos W., Seilmaier M., Zange S., Müller M.A. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020;581:465–469. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

