Protective effects of VMY-2-95 on corticosterone-induced injuries in mice and cellular models
- PMID: 34386327
- PMCID: PMC8343195
- DOI: 10.1016/j.apsb.2021.03.002
Protective effects of VMY-2-95 on corticosterone-induced injuries in mice and cellular models
Erratum in
-
Erratum: Author correction to 'Protective effects of VMY-2-95 on corticosterone-induced injuries in mice and cellular models' [Acta Pharmaceutica Sinica B 11(2021) 1903-1913].Acta Pharm Sin B. 2023 Feb;13(2):899-901. doi: 10.1016/j.apsb.2022.11.017. Epub 2022 Nov 17. Acta Pharm Sin B. 2023. PMID: 36873189 Free PMC article.
-
Erratum: Author correction to 'Protective effects of VMY-2-95 on corticosterone-induced injuries in mice and cellular models' [Acta Pharmaceutica Sinica B 11 (2021) 1903-1913].Acta Pharm Sin B. 2023 Aug;13(8):3579-3580. doi: 10.1016/j.apsb.2023.05.002. Epub 2023 May 7. Acta Pharm Sin B. 2023. PMID: 37655326 Free PMC article.
Abstract
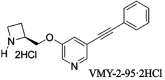
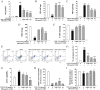
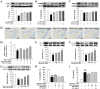
Nicotinic α4β2 receptor antagonists have drawn increasing attention in the development of new antidepressants. In this study, we aimed to investigate the protective effect of VMY-2-95, the new selective antagonist of α4β2 nicotinic acetylcholine receptor (nAChR) on corticosterone (CORT) injured mice and cellular models. Fluoxetine was applied as a positive control, and the effects of VMY-2-95 were investigated with three different doses or concentrations (1, 3, 10 mg/kg in mice, and 0.003, 0.03, 0.1 μmol/L in cells). As a result, VMY-2-95 showed significant antidepressant-like effects in the CORT injured mice by improving neuromorphic function, promoting hippocampal nerve proliferation, and regulating the contents of monoamine transmitters. Meanwhile, VMY-2-95 exhibited protective effects on cell viability, cell oxidant, cell apoptosis, and mitochondrial energy metabolism on corticosterone-impaired SH-SY5Y cells. Also, the PKA-CREB-BDNF signaling pathway was up-regulated by VMY-2-95 both in vitro and in vivo, and pathway blockers were also combined with VMY-2-95 to verify the effects furtherly. Therefore, we preliminarily proved that VMY-2-95 had protective effects in depressed mice and SH-SY5Y cells against injuries induced by corticosterone. This work indicated that the application of VMY-2-95 is a potential pharmacological solution for depression. This study also supported the development of α4β2 nAChR antagonists towards neuropsychiatric dysfunctions.
Keywords: Cholinergic–adrenergic theory; Corticosterone; Depression; PKA-CREB-BDNF signaling pathway; SH-SY5Y cells; VMY-2-95; α4β2 nAChR antagonist.
© 2021 Chinese Pharmaceutical Association and Institute of Materia Medica, Chinese Academy of Medical Sciences. Production and hosting by Elsevier B.V.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Preclinical studies of the potent and selective nicotinic α4β2 receptor ligand VMY-2-95.Mol Pharm. 2015 Feb 2;12(2):393-402. doi: 10.1021/mp5003569. Epub 2015 Jan 20. Mol Pharm. 2015. PMID: 25533629 Free PMC article.
-
Involvement of PI3K/Akt/FoxO3a and PKA/CREB Signaling Pathways in the Protective Effect of Fluoxetine Against Corticosterone-Induced Cytotoxicity in PC12 Cells.J Mol Neurosci. 2016 Aug;59(4):567-78. doi: 10.1007/s12031-016-0779-7. Epub 2016 Jul 13. J Mol Neurosci. 2016. PMID: 27412469
-
2-Phenylethylamine (PEA) Ameliorates Corticosterone-Induced Depression-Like Phenotype via the BDNF/TrkB/CREB Signaling Pathway.Int J Mol Sci. 2020 Nov 30;21(23):9103. doi: 10.3390/ijms21239103. Int J Mol Sci. 2020. PMID: 33265983 Free PMC article.
-
The contribution of agonist and antagonist activities of α4β2* nAChR ligands to smoking cessation efficacy: a quantitative analysis of literature data.Psychopharmacology (Berl). 2018 Sep;235(9):2479-2505. doi: 10.1007/s00213-018-4921-9. Epub 2018 Jul 7. Psychopharmacology (Berl). 2018. PMID: 29980822 Review.
-
Advances in the In vitro and In vivo pharmacology of Alpha4beta2 nicotinic receptor positive allosteric modulators.Neuropharmacology. 2020 May 15;168:108008. doi: 10.1016/j.neuropharm.2020.108008. Epub 2020 Feb 12. Neuropharmacology. 2020. PMID: 32113032 Free PMC article. Review.
Cited by
-
Essential oil from the roots of Paeonia lactiflora pall. has protective effect against corticosterone-induced depression in mice via modulation of PI3K/Akt signaling pathway.Front Pharmacol. 2022 Sep 16;13:999712. doi: 10.3389/fphar.2022.999712. eCollection 2022. Front Pharmacol. 2022. PMID: 36188568 Free PMC article.
-
Serotonin Type 3 Receptor Is Potentially Involved in Cellular Stress Induced by Hydrogen Peroxide.Life (Basel). 2022 Oct 19;12(10):1645. doi: 10.3390/life12101645. Life (Basel). 2022. PMID: 36295079 Free PMC article.
-
High-throughput discovery of highly selective reversible hMAO-B inhibitors based on at-line nanofractionation.Acta Pharm Sin B. 2024 Apr;14(4):1772-1786. doi: 10.1016/j.apsb.2024.01.020. Epub 2024 Feb 6. Acta Pharm Sin B. 2024. PMID: 38572096 Free PMC article.
-
Revealing the Mechanism of Hemerocallis citrina Baroni in Depression Treatment Through Integrated Network Pharmacology and Transcriptomic Analysis.Pharmaceuticals (Basel). 2024 Dec 17;17(12):1704. doi: 10.3390/ph17121704. Pharmaceuticals (Basel). 2024. PMID: 39770546 Free PMC article.
-
Reduction in SH-SY5Y Cell Stress Induced by Corticosterone and Attenuation of the Inflammatory Response in RAW 264.7 Cells Using Endomorphin Analogs.Biomedicines. 2025 Jul 20;13(7):1774. doi: 10.3390/biomedicines13071774. Biomedicines. 2025. PMID: 40722844 Free PMC article.
References
-
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390:1211–1259. - PMC - PubMed
-
- World Health Organization (WHO). Depression. World Health Organization. Published [February 2017]. Updated [30 January 2020]. Accessed [25 June, 2019]. Available from: https://www.who.int/en/news-room/fact-sheets/detail/depression.
-
- Chang T., Fava M. The future of psychopharmacology of depression. J Clin Psychiatry. 2010;71:971–975. - PubMed
-
- Janowsky D.S., el-Yousef M.K., Davis J.M., Sekerke H.J. A cholinergic-adrenergic hypothesis of mania and depression. Lancet. 1972;2:632–635. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

