Protein cofactors and substrate influence Mg2+-dependent structural changes in the catalytic RNA of archaeal RNase P
- PMID: 34387688
- PMCID: PMC8450104
- DOI: 10.1093/nar/gkab655
Protein cofactors and substrate influence Mg2+-dependent structural changes in the catalytic RNA of archaeal RNase P
Abstract
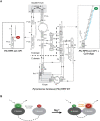
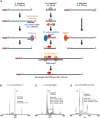
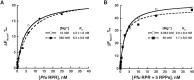
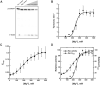
The ribonucleoprotein (RNP) form of archaeal RNase P comprises one catalytic RNA and five protein cofactors. To catalyze Mg2+-dependent cleavage of the 5' leader from pre-tRNAs, the catalytic (C) and specificity (S) domains of the RNase P RNA (RPR) cooperate to recognize different parts of the pre-tRNA. While ∼250-500 mM Mg2+ renders the archaeal RPR active without RNase P proteins (RPPs), addition of all RPPs lowers the Mg2+ requirement to ∼10-20 mM and improves the rate and fidelity of cleavage. To understand the Mg2+- and RPP-dependent structural changes that increase activity, we used pre-tRNA cleavage and ensemble FRET assays to characterize inter-domain interactions in Pyrococcus furiosus (Pfu) RPR, either alone or with RPPs ± pre-tRNA. Following splint ligation to doubly label the RPR (Cy3-RPRC domain and Cy5-RPRS domain), we used native mass spectrometry to verify the final product. We found that FRET correlates closely with activity, the Pfu RPR and RNase P holoenzyme (RPR + 5 RPPs) traverse different Mg2+-dependent paths to converge on similar functional states, and binding of the pre-tRNA by the holoenzyme influences Mg2+ cooperativity. Our findings highlight how Mg2+ and proteins in multi-subunit RNPs together favor RNA conformations in a dynamic ensemble for functional gains.
© The Author(s) 2021. Published by Oxford University Press on behalf of Nucleic Acids Research.
Figures

Similar articles
-
Cooperative RNP assembly: complementary rescue of structural defects by protein and RNA subunits of archaeal RNase P.J Mol Biol. 2011 Aug 12;411(2):368-83. doi: 10.1016/j.jmb.2011.05.012. Epub 2011 Jun 12. J Mol Biol. 2011. PMID: 21683084 Free PMC article.
-
Cleavage of model substrates by archaeal RNase P: role of protein cofactors in cleavage-site selection.Nucleic Acids Res. 2011 Feb;39(3):1105-16. doi: 10.1093/nar/gkq732. Epub 2010 Oct 8. Nucleic Acids Res. 2011. PMID: 20935047 Free PMC article.
-
Functional reconstitution and characterization of Pyrococcus furiosus RNase P.Proc Natl Acad Sci U S A. 2006 Oct 31;103(44):16147-52. doi: 10.1073/pnas.0608000103. Epub 2006 Oct 19. Proc Natl Acad Sci U S A. 2006. PMID: 17053064 Free PMC article.
-
Structural basis for activation of an archaeal ribonuclease P RNA by protein cofactors.Biosci Biotechnol Biochem. 2017 Sep;81(9):1670-1680. doi: 10.1080/09168451.2017.1353404. Epub 2017 Jul 17. Biosci Biotechnol Biochem. 2017. PMID: 28715256 Review.
-
The Dynamic Network of RNP RNase P Subunits.Int J Mol Sci. 2021 Sep 24;22(19):10307. doi: 10.3390/ijms221910307. Int J Mol Sci. 2021. PMID: 34638646 Free PMC article. Review.
Cited by
-
The many faces of RNA-based RNase P, an RNA-world relic.Trends Biochem Sci. 2021 Dec;46(12):976-991. doi: 10.1016/j.tibs.2021.07.005. Epub 2021 Sep 9. Trends Biochem Sci. 2021. PMID: 34511335 Free PMC article. Review.
-
Use of a small molecule microarray screen to identify inhibitors of the catalytic RNA subunit of Methanobrevibacter smithii RNase P.Nucleic Acids Res. 2025 Jan 7;53(1):gkae1190. doi: 10.1093/nar/gkae1190. Nucleic Acids Res. 2025. PMID: 39676671 Free PMC article.
-
Extremophiles: the species that evolve and survive under hostile conditions.3 Biotech. 2023 Sep;13(9):316. doi: 10.1007/s13205-023-03733-6. Epub 2023 Aug 25. 3 Biotech. 2023. PMID: 37637002 Free PMC article. Review.
-
Divergent molecular assembly and catalytic mechanisms between bacterial and archaeal RNase P in pre-tRNA cleavage.Proc Natl Acad Sci U S A. 2024 Oct 22;121(43):e2407579121. doi: 10.1073/pnas.2407579121. Epub 2024 Oct 16. Proc Natl Acad Sci U S A. 2024. PMID: 39413135 Free PMC article.
-
Elucidation of structure-function relationships in Methanocaldococcus jannaschii RNase P, a multi-subunit catalytic ribonucleoprotein.Nucleic Acids Res. 2022 Aug 12;50(14):8154-8167. doi: 10.1093/nar/gkac595. Nucleic Acids Res. 2022. PMID: 35848927 Free PMC article.
References
-
- Froschauer E.M., Kolisek M., Dieterich F., Schweigel M., Schweyen R.J.. Fluorescence measurements of free [Mg2+] by use of mag-fura 2 in Salmonella enterica. FEMS Microbiol. Lett. 2004; 237:49–55. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

