The Evolution of Single-Cell Analysis and Utility in Drug Development
- PMID: 34389904
- PMCID: PMC8363238
- DOI: 10.1208/s12248-021-00633-6
The Evolution of Single-Cell Analysis and Utility in Drug Development
Abstract
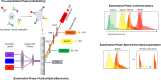
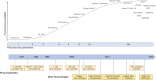
This review provides a brief history of the advances of cellular analysis tools focusing on instrumentation, detection probes, and data analysis tools. The interplay of technological advancement and a deeper understanding of cellular biology are emphasized. The relevance of this topic to drug development is that the evaluation of cellular biomarkers has become a critical component of the development strategy for novel immune therapies, cell therapies, gene therapies, antiviral therapies, and vaccines. Moreover, recent technological advances in single-cell analysis are providing more robust cellular measurements and thus accelerating the advancement of novel therapies.Graphical abstract.
Keywords: drug development; flow cytometry; single cell.
© 2021. American Association of Pharmaceutical Scientists.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Single cell analyses of development in the modern era.Development. 2019 Jun 27;146(12):dev181396. doi: 10.1242/dev.181396. Development. 2019. PMID: 31249004 No abstract available.
-
Reimagining Flow Cytometric Cell Sorting.Adv Biosyst. 2020 Aug;4(8):e2000019. doi: 10.1002/adbi.202000019. Epub 2020 Jun 2. Adv Biosyst. 2020. PMID: 32483933 Review.
-
Cell motility under the microscope: Vorsprung durch Technik.Nat Rev Mol Cell Biol. 2004 Aug;5(8):667-72. doi: 10.1038/nrm1439. Nat Rev Mol Cell Biol. 2004. PMID: 15366710 Review.
-
Mass cytometry and type 1 diabetes research in the age of single-cell data science.Curr Opin Endocrinol Diabetes Obes. 2020 Aug;27(4):231-239. doi: 10.1097/MED.0000000000000549. Curr Opin Endocrinol Diabetes Obes. 2020. PMID: 32618635 Free PMC article. Review.
-
The new era of quantitative cell imaging-challenges and opportunities.Mol Cell. 2022 Jan 20;82(2):241-247. doi: 10.1016/j.molcel.2021.12.024. Mol Cell. 2022. PMID: 35063094 Free PMC article. Review.
Cited by
-
Bioanalytical Assay Strategies and Considerations for Measuring Cellular Kinetics.Int J Mol Sci. 2022 Dec 31;24(1):695. doi: 10.3390/ijms24010695. Int J Mol Sci. 2022. PMID: 36614138 Free PMC article. Review.
-
Using Spectral Flow Cytometry for CAR T-Cell Clinical Trials: Game Changing Technologies Enabling Novel Therapies.Int J Mol Sci. 2024 Sep 24;25(19):10263. doi: 10.3390/ijms251910263. Int J Mol Sci. 2024. PMID: 39408593 Free PMC article. Review.
-
OMIP-095: 40-Color spectral flow cytometry delineates all major leukocyte populations in murine lymphoid tissues.Cytometry A. 2023 Nov;103(11):839-850. doi: 10.1002/cyto.a.24788. Epub 2023 Sep 28. Cytometry A. 2023. PMID: 37768325 Free PMC article.
-
Full spectrum flow cytometry and mass cytometry: A 32-marker panel comparison.Cytometry A. 2022 Nov;101(11):942-959. doi: 10.1002/cyto.a.24565. Epub 2022 May 20. Cytometry A. 2022. PMID: 35593221 Free PMC article.
-
2024 White paper on recent issues in bioanalysis: Impact of LDT in US and IVDR in EU; AI/ML for High Parameter Flow Cytometry; The rise of Olink Technology; CDx for AAV Gene Therapies; Integrative Bioanalysis by Multiple Platforms; Super Sensitive ADA/NAb LBA (PART 2A - Recommendations on Advanced Strategies for Biomarkers, IVD/CDx Assays (BAV), Cell Based Assays (CBA), and Ligand-Binding Assays (LBA) PART 2B - Regulatory Agencies' Input on Biomarkers, IVD/CDx, and Biomarker Assay Validation).Bioanalysis. 2025 Feb;17(4):211-248. doi: 10.1080/17576180.2024.2442218. Epub 2025 Jan 25. Bioanalysis. 2025. PMID: 39862107
References
-
- Raventos-Suarez C, Long B. A multiparameter approach to cell cycle analysis as a standard tool in oncology drug discovery. In: Litwin V, Marder P, editors. Flow Cytometry in Drug Discovery and Development. Wiley-Blackwell, John Wiley & Sons, Inc.: New Jersey; 2011. pp. 99–122.
-
- Sarikonda G, Mathieu M, Natalia M, Pahuja A, Xue Q, Pierog PL, et al. Best practices for the development, analytical validation and clinical implementation of flow cytometric methods for chimeric antigen receptor T cell analyses. Cytom Part B - Clin Cytom [Internet]. 2021 Jan 1 [cited 2021 Mar 19];100(1):79–91. Available from: https://pubmed.ncbi.nlm.nih.gov/33373096/ - PubMed
-
- Sarikonda G, Pahuja A, Kalfoglou C, Burns K, Nguyen K, Ch’en IL, et al. Monitoring CAR-T cell kinetics in clinical trials by multiparametric flow cytometry: benefits and challenges [Internet]. Vol. 100, Cytometry Part B - Clinical Cytometry. John Wiley and Sons Inc; 2021 [cited 2021 Mar 19]. p. 72–8. Available from: https://pubmed.ncbi.nlm.nih.gov/32573972/ - PubMed
-
- Stewart JJ, Green CL, Jones N, Liang M, Xu Y, Wilkins DEC, et al. Role of receptor occupancy assays by flow cytometry in drug development [Internet]. Vol. 90, Cytometry. Part B, Clinical cytometry. Cytometry B Clin Cytom; 2016 [cited 2021 Mar 19]. p. 110–6. Available from: https://pubmed.ncbi.nlm.nih.gov/26704557/ - PubMed
-
- Green CL, Stewart JJ, Högerkorp CM, Lackey A, Jones N, Liang M, et al. Recommendations for the development and validation of flow cytometry-based receptor occupancy assays [Internet]. Vol. 90, Cytometry. Part B, Clinical cytometry. Cytometry B Clin Cytom; 2016 [cited 2021 Mar 19]. p. 141–9. Available from: https://pubmed.ncbi.nlm.nih.gov/26566147/ - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

