TGFB-induced factor homeobox 1 (TGIF) expression in breast cancer
- PMID: 34391399
- PMCID: PMC8364691
- DOI: 10.1186/s12885-021-08656-0
TGFB-induced factor homeobox 1 (TGIF) expression in breast cancer
Erratum in
-
Correction to: TGFB-induced factor homeobox 1 (TGIF) expression in breast cancer.BMC Cancer. 2021 Sep 15;21(1):1024. doi: 10.1186/s12885-021-08754-z. BMC Cancer. 2021. PMID: 34526003 Free PMC article. No abstract available.
Abstract
Background: Breast cancer (BC) is the most frequent female cancer and preferentially metastasizes to bone. The transcription factor TGFB-induced factor homeobox 1 (TGIF) is involved in bone metabolism. However, it is not yet known whether TGIF is associated with BC bone metastasis or patient outcome and thus of potential interest.
Methods: TGIF expression was analyzed by immunohistochemistry in 1197 formalin-fixed, paraffin-embedded tissue samples from BC patients treated in the GAIN (German Adjuvant Intergroup Node-Positive) study with two adjuvant dose-dense schedules of chemotherapy with or without bisphosphonate ibandronate. TGIF expression was categorized into negative/low and moderate/strong staining. Endpoints were disease-free survival (DFS), overall survival (OS) and time to primary bone metastasis as first site of relapse (TTPBM).
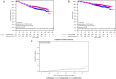
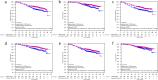
Results: We found associations of higher TGIF protein expression with smaller tumor size (p = 0.015), well differentiated phenotype (p < 0.001) and estrogen receptor (ER)-positive BC (p < 0.001). Patients with higher TGIF expression levels showed a significantly longer disease-free (DFS: HR 0.75 [95%CI 0.59-0.95], log-rank p = 0.019) and overall survival (OS: HR 0.69 [95%CI 0.50-0.94], log-rank p = 0.019), but no association with TTPBM (HR 0.77 [95%CI 0.51-1.16]; p = 0.213). Univariate analysis in molecular subgroups emphasized that elevated TGIF expression was prognostic for both DFS and OS in ER-positive BC patients (DFS: HR 0.68 [95%CI 0.51-0.91]; log-rank p = 0.009, interaction p = 0.130; OS: HR 0.60 [95%CI 0.41-0.88], log-rank p = 0.008, interaction p = 0.107) and in the HER2-negative subgroup (DFS:HR 0.67 [95%CI 0.50-0.88], log-rank p = 0.004, interaction p = 0.034; OS: HR 0.57 [95%CI 0.40-0.81], log-rank p = 0.002, interaction p = 0.015).
Conclusions: Our results suggest that moderate to high TGIF expression is a common feature of breast cancer cells and that this is not associated with bone metastases as first site of relapse. However, a reduced expression is linked to tumor progression, especially in HER2-negative breast cancer.
Trial registration: This clinical trial has been registered with ClinicalTrials.gov ; registration number: NCT00196872 .
Keywords: Bone metastases; Breast cancer; TGFB-induced factor homeobox 1; TGIF.
© 2021. The Author(s).
Conflict of interest statement
V.M: Dr. Möbus received speaker honoraria from Amgen, Astra Zeneca, Celgene, Roche, Teva, and consultancy honoraria from Roche, Amgen, Tesaro and Myelo Therapeutics GmbH.
V.M: Dr. Müller received speaker honoraria from Amgen, Astra Zeneca, Celgene, Daiichi-Sankyo, Eisai, Pfizer, Novartis, Roche, Teva, and consultancy honoraria from Genomic Health, Hexal, Roche, Pierre Fabre, Amgen, Novartis, MSD, Daiichi-Sankyo and Eisai, Lilly, Tesaro and Nektar. Research support from Novartis, Roche, Seattle Genetics, Genentech.
PAF: Dr. Fasching reports grants from Novartis, grants from Biontech, personal fees from Novartis, personal fees from Roche, personal fees from Pfizer, personal fees from Celgene, personal fees from Daiichi-Sankyo, personal fees from Astra Zeneca, personal fees from Macrogenics, personal fees from Eisai, personal fees from Merck Sharp & Dohme, grants from Cepheid, personal fees from Lilly, during the conduct of the study.
ES: Dr. Stickeler reports personal fees from Roche Pharma, personal fees from Novartis, personal fees from Pfizer, personal fees from Tesaro, personal fees from Astra Zeneca, outside the submitted work.
CD: Dr. Denkert received honoraria from Novartis and Roche; Research funding from Myriad Genetics, reports consulting or advisory role for MSD Oncology and Daiichi Sankyo and stock and other ownership interest with Sividon Diagnostics (now Myriad and travel expenses from Roche. In addition, Dr. Denkert has patents, royalities and intellectual property with VMscopedigital pathology software: Patent application: EP18209672 - cancer immunotherapy; Patent application EP20150702464 - therapy response; Patent application EP20150702464 - therapy response.
LH: Dr. Hanker received speaker honoraria from Astra Zeneca, Clovis, GSK/Tesaro Novartis, Roche, and consultancy honoraria from Astra Zeneca, Clovis, GSK/Tesaro and Roche.
FM: Dr. Marmé reports personal fees from Roche, personal fees from AstraZeneca, personal fees from Pfizer, personal fees from Tesaro, personal fees from Novartis, personal fees from Amgen, personal fees from PharmaMar, personal fees from GenomicHealth, personal fees from CureVac, personal fees from EISAI, outside the submitted work.
SL: Dr. Loibl reports grants and other from Abbvie, grants and other from Amgen, grants and other from Astra Zeneca, grants and other from Celgene, grants and other from Novartis, grants and other from Pfizer, grants and other from Roche, other from Seattle Genetics, other from PriME/ Medscape, personal fees from Chugai, grants from Teva, grants from Vifor, grants and other from Daiichi-Sankyo, other from Lilly, other from Samsung, other from Eirgenix, other from BMS, other from Puma, other from MSD, grants from Immunomedics, outside the submitted work; In addition, Dr. Loibl has a patent EP14153692.0 pending.
The other authors (CS; KML; SS; JR; RPH; TK; CS; CHK; LH; US; VV; VN) declare that they have no competing interests.
Figures



References
-
- Menshawy A, Mattar O, Abdulkarim A, Kasem S, Nasreldin N, Menshawy E, et al. Denosumab versus bisphosphonates in patients with advanced cancers-related bone metastasis: systematic review and meta-analysis of randomized controlled trials. Support Care Cancer. 2018;26(4):1029–38. 10.1007/s00520-018-4060-1. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

