Trialkyl(vinyl)phosphonium Chlorophenol Derivatives as Potent Mitochondrial Uncouplers and Antibacterial Agents
- PMID: 34396013
- PMCID: PMC8359139
- DOI: 10.1021/acsomega.1c02909
Trialkyl(vinyl)phosphonium Chlorophenol Derivatives as Potent Mitochondrial Uncouplers and Antibacterial Agents
Abstract
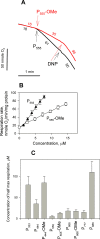
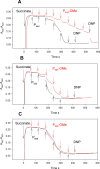
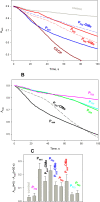
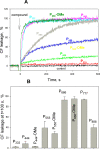
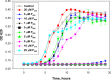
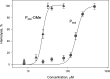
Trialkyl phosphonium derivatives of vinyl-substituted p-chlorophenol were synthesized here by a recently developed method of preparing quaternary phosphonium salts from phosphine oxides using Grignard reagents. All the derivatives with a number (n) of carbon atoms in phosphonium alkyl substituents varying from 4 to 7 showed pronounced uncoupling activity in isolated rat liver mitochondria at micromolar concentrations, with a tripentyl derivative being the most effective both in accelerating respiration and causing membrane potential collapse, as well as in provoking mitochondrial swelling in a potassium-acetate medium. Remarkably, the trialkyl phosphonium derivatives with n from 4 to 7 also proved to be rather potent antibacterial agents. Methylation of the chlorophenol hydroxyl group suppressed the effects of P555 and P444 on the respiration and membrane potential of mitochondria but not those of P666, thereby suggesting a mechanistic difference in the mitochondrial uncoupling by these derivatives, which was predominantly protonophoric (carrier-like) in the case of P555 and P444 but detergent-like with P666. The latter was confirmed by the carboxyfluorescein leakage assay on model liposomal membranes.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
-
- Antonenko Y. N.; Avetisyan A. V.; Bakeeva L. E.; Chernyak B. V.; Chertkov V. A.; Domnina L. V.; Ivanova O. Y.; Izyumov D. S.; Khailova L. S.; Klishin S. S.; Korshunova G. A.; Lyamzaev K. G.; Muntyan M. S.; Nepryakhina O. K.; Pashkovskaya A. A.; Pletjushkina O. Y.; Pustovidko A. V.; Roginsky V. A.; Rokitskaya T. I.; Ruuge E. K.; Saprunova V. B.; Severina I. I.; Simonyan R. A.; Skulachev I. V.; Skulachev M. V.; Sumbatyan N. V.; Sviryaeva I. V.; Tashlitsky V. N.; Vassiliev J. M.; Vyssokikh M. Y.; Yaguzhinsky L. S.; Zamyatnin A. A. Jr.; Skulachev V. P. Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 1. Cationic plastoquinone derivatives: synthesis and in vitro studies. Biochemistry (Moscow) 2008, 73, 1273–1287. 10.1134/S0006297908120018. - DOI - PubMed
LinkOut - more resources
Full Text Sources

