Bioengineered optogenetic model of human neuromuscular junction
- PMID: 34403849
- PMCID: PMC8439334
- DOI: 10.1016/j.biomaterials.2021.121033
Bioengineered optogenetic model of human neuromuscular junction
Abstract
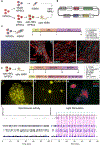
Functional human tissues engineered from patient-specific induced pluripotent stem cells (hiPSCs) hold great promise for investigating the progression, mechanisms, and treatment of musculoskeletal diseases in a controlled and systematic manner. For example, bioengineered models of innervated human skeletal muscle could be used to identify novel therapeutic targets and treatments for patients with complex central and peripheral nervous system disorders. There is a need to develop standardized and objective quantitative methods for engineering and using these complex tissues, in order increase their robustness, reproducibility, and predictiveness across users. Here we describe a standardized method for engineering an isogenic, patient specific human neuromuscular junction (NMJ) that allows for automated quantification of NMJ function to diagnose disease using a small sample of blood serum and evaluate new therapeutic modalities. By combining tissue engineering, optogenetics, microfabrication, optoelectronics and video processing, we created a novel platform for the precise investigation of the development and degeneration of human NMJ. We demonstrate the utility of this platform for the detection and diagnosis of myasthenia gravis, an antibody-mediated autoimmune disease that disrupts the NMJ function.
Keywords: Disease modeling; Human tissue models; Myasthenia gravis; Neuromuscular junction; Optogenetics; iPS cells.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
CONFLICTS OF INTEREST
The authors declare no conflict of interest.
Declaration of interests
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:
GVN is a co-founder of Tara Biosystems, a Columbia University start-up that is commercializing “heart on a chip” platform for drug discovery. She holds equity, serves on the Board of Directors, is an inventor on licensed by Tara, and has received financial compensation.
Figures





References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

