Potential of Autologous Progenitor Cells and Decellularized Porcine Artery Matrix in Construction of Tissue-engineered Vascular Grafts
- PMID: 34405770
- PMCID: PMC9208767
- DOI: 10.1080/15476278.2021.1963603
Potential of Autologous Progenitor Cells and Decellularized Porcine Artery Matrix in Construction of Tissue-engineered Vascular Grafts
Abstract
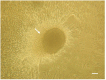
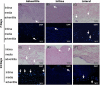
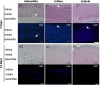
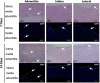
To develop a tissue-engineered vascular graft, we used pericardial effusion-derived progenitor cells (PEPCs) collected from drained fluid after open-heart surgery in children with congenital heart diseases to repopulate a decellularized porcine pulmonary artery. The PEPCs were compared with human fibroblasts (HS68) and human umbilical vein endothelial cells (HUVECs) in cell growth and migration. They were cultured with the matrices via an inner approach (intima), lateral approach (media), and outer approach (adventitia). PEPCs grew and migrated better than the other two cells 14 days after seeding in the decellularized vessel. In immunofluorescence assays, PEPCs expressed CD90 and CD105 indicating a vascular differentiation. PEPCs grew in a decellularized porcine pulmonary artery matrix may have the potential for producing tissue-engineered vascular grafts.
Keywords: Tissue-engineered vascular graft; decellularized biological scaffolds; pericardial effuse drain fluid; progenitor cells.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures




Similar articles
-
Construction of tissue-engineered heart valves by using decellularized scaffolds and endothelial progenitor cells.Chin Med J (Engl). 2007 Apr 20;120(8):696-702. Chin Med J (Engl). 2007. PMID: 17517187
-
Decellularized tissue-engineered blood vessel as an arterial conduit.Proc Natl Acad Sci U S A. 2011 May 31;108(22):9214-9. doi: 10.1073/pnas.1019506108. Epub 2011 May 12. Proc Natl Acad Sci U S A. 2011. PMID: 21571635 Free PMC article.
-
[Preliminary in vivo evaluation of tissue engineered venous grafts fabricated based on endothelial progenitor cells].Zhonghua Wai Ke Za Zhi. 2007 Apr 1;45(7):491-5. Zhonghua Wai Ke Za Zhi. 2007. PMID: 17686312 Chinese.
-
Challenges and Strategies for Endothelializing Decellularized Small-Diameter Tissue-Engineered Vessel Grafts.Adv Healthc Mater. 2024 Jun;13(16):e2304432. doi: 10.1002/adhm.202304432. Epub 2024 Mar 17. Adv Healthc Mater. 2024. PMID: 38462702 Review.
-
Scaffolds in tissue engineering of blood vessels.Can J Physiol Pharmacol. 2010 Sep;88(9):855-73. doi: 10.1139/y10-073. Can J Physiol Pharmacol. 2010. PMID: 20921972 Review.
Cited by
-
Mitigating challenges and expanding the future of vascular tissue engineering-are we there yet?Front Physiol. 2023 Jan 4;13:1079421. doi: 10.3389/fphys.2022.1079421. eCollection 2022. Front Physiol. 2023. PMID: 36685187 Free PMC article. No abstract available.
-
Strategies to counteract adverse remodeling of vascular graft: A 3D view of current graft innovations.Front Bioeng Biotechnol. 2023 Jan 10;10:1097334. doi: 10.3389/fbioe.2022.1097334. eCollection 2022. Front Bioeng Biotechnol. 2023. PMID: 36704297 Free PMC article. Review.
-
Biological Scaffolds for Congenital Heart Disease.Bioengineering (Basel). 2023 Jan 2;10(1):57. doi: 10.3390/bioengineering10010057. Bioengineering (Basel). 2023. PMID: 36671629 Free PMC article. Review.
-
Fabrication of a Triple-Layer Bionic Vascular Scaffold via Hybrid Electrospinning.J Funct Biomater. 2024 May 23;15(6):140. doi: 10.3390/jfb15060140. J Funct Biomater. 2024. PMID: 38921514 Free PMC article.
References
-
- Deaths: HM. Leading causes for 2016 national vital statistics reports: from the centers for disease control and prevention, national center for health statistics. National Vital Statistics System. 2018;67:1–77. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
