SARS-CoV-2 natural infection in animals: a systematic review of studies and case reports and series
- PMID: 34406913
- PMCID: PMC8428274
- DOI: 10.1080/01652176.2021.1970280
SARS-CoV-2 natural infection in animals: a systematic review of studies and case reports and series
Abstract
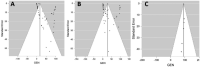
COVID-19 pandemic is essentially a zoonotic disease. In this context, early in 2020, transmission from humans to certain animals began reporting; the number of studies has grown since. To estimate the pooled prevalence of SARS-CoV-2 natural infection in animals and to determine differences in prevalence between countries, years, animal types and diagnostic methods (RT-PCR or serological tests). A systematic literature review with meta-analysis using eight databases. Observational studies were included but analyzed separately. We performed a random-effects model meta-analysis to calculate the pooled prevalence and 95% confidence interval (95% CI) for prevalence studies and case series. After the screening, 65 reports were selected for full-text assessment and included for qualitative and quantitative analyses. A total of 24 reports assessed SARS-CoV-2 infection by RT-PCR, combining a total of 321,785 animals, yielding a pooled prevalence of 12.3% (95% CI 11.6%-13.0%). Also, a total of 17 studies additionally assessed serological response against SARS-CoV-2, including nine by ELISA, four by PRTN, one by MIA, one by immunochromatography (rest, two studies, the method was not specified), combining a total of 5319 animals, yielding a pooled prevalence of 29.4% (95% CI 22.9%-35.9%). A considerable proportion of animals resulted infected by SARS-CoV-2, ranking minks among the highest value, followed by dogs and cats. Further studies in other animals are required to define the extent and importance of natural infection due to SARS-CoV-2. These findings have multiple implications for public human and animal health. One Health approach in this context is critical for prevention and control.
Keywords: COVID-19; SARS-CoV-2; animals; prevalence; transmission; zoonotic.
Conflict of interest statement
All authors report no potential conflicts.
Figures










References
-
- Bartlett SL, Diel DG, Wang L, Zec S, Laverack M, Martins M, Caserta LC, Killian ML, Terio K, Olmstead C, et al. 2021. SARS-COV-2 infection and longitudinal fecal screening in Malayan Tigers (Panthera Tigris Jacksoni), Amur Tigers (Panthera Tigris Altaica), and African Lions (Panthera Leo Krugeri) at the Bronx Zoo, New York, USA. J Zoo Wildl Med. 51(4):733–744. - PubMed
-
- Bonilla-Aldana DK, Dhama K, Rodriguez-Morales AJ.. 2020. Revisiting the One Health approach in the context of COVID-19: a look into the ecology of this emerging disease. Adv Anim Vet Sci. 8:234–237.
-
- Bonilla-Aldana DK, Jimenez-Diaz SD, Arango-Duque JS, Aguirre-Florez M, Balbin-Ramon GJ, Paniz-Mondolfi A, Suarez JA, Pachar MR, Perez-Garcia LA, Delgado-Noguera LA, et al. 2021. Bats in ecosystems and their Wide spectrum of viral infectious potential threats: SARS-CoV-2 and other emerging viruses. Int J Infect Dis. 102:87–96. - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
