Insights into the bilayer-mediated toppling mechanism of a folate-specific ECF transporter by cryo-EM
- PMID: 34408021
- PMCID: PMC8403918
- DOI: 10.1073/pnas.2105014118
Insights into the bilayer-mediated toppling mechanism of a folate-specific ECF transporter by cryo-EM
Abstract
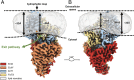
Energy-coupling factor (ECF)-type transporters are small, asymmetric membrane protein complexes (∼115 kDa) that consist of a membrane-embedded, substrate-binding protein (S component) and a tripartite ATP-hydrolyzing module (ECF module). They import micronutrients into bacterial cells and have been proposed to use a highly unusual transport mechanism, in which the substrate is dragged across the membrane by a toppling motion of the S component. However, it remains unclear how the lipid bilayer could accommodate such a movement. Here, we used cryogenic electron microscopy at 200 kV to determine structures of a folate-specific ECF transporter in lipid nanodiscs and detergent micelles at 2.7- and 3.4-Å resolution, respectively. The structures reveal an irregularly shaped bilayer environment around the membrane-embedded complex and suggest that toppling of the S component is facilitated by protein-induced membrane deformations. In this way, structural remodeling of the lipid bilayer environment is exploited to guide the transport process.
Keywords: ABC transporter; cryo-EM; lipid bilayer deformation; membrane transport.
Conflict of interest statement
The authors declare no competing interest.
Figures



Similar articles
-
Membrane mediated toppling mechanism of the folate energy coupling factor transporter.Nat Commun. 2020 Apr 9;11(1):1763. doi: 10.1038/s41467-020-15554-9. Nat Commun. 2020. PMID: 32273501 Free PMC article.
-
Structural insight in the toppling mechanism of an energy-coupling factor transporter.Nat Commun. 2016 Mar 30;7:11072. doi: 10.1038/ncomms11072. Nat Commun. 2016. PMID: 27026363 Free PMC article.
-
Functional and structural characterization of an ECF-type ABC transporter for vitamin B12.Elife. 2018 May 29;7:e35828. doi: 10.7554/eLife.35828. Elife. 2018. PMID: 29809140 Free PMC article.
-
ECF-Type ATP-Binding Cassette Transporters.Annu Rev Biochem. 2019 Jun 20;88:551-576. doi: 10.1146/annurev-biochem-013118-111705. Epub 2019 Nov 28. Annu Rev Biochem. 2019. PMID: 30485755 Review.
-
Energy coupling factor-type ABC transporters for vitamin uptake in prokaryotes.Biochemistry. 2012 Jun 5;51(22):4390-6. doi: 10.1021/bi300504v. Epub 2012 May 21. Biochemistry. 2012. PMID: 22574898 Review.
Cited by
-
Identification of inhibitors targeting the energy-coupling factor (ECF) transporters.Commun Biol. 2023 Nov 20;6(1):1182. doi: 10.1038/s42003-023-05555-x. Commun Biol. 2023. PMID: 37985798 Free PMC article.
-
MreC-MreD structure reveals a multifaceted interface that controls MreC conformation.bioRxiv [Preprint]. 2024 Oct 8:2024.10.08.617240. doi: 10.1101/2024.10.08.617240. bioRxiv. 2024. PMID: 39416049 Free PMC article. Preprint.
-
Bidirectional ATP-driven transport of cobalamin by the mycobacterial ABC transporter BacA.Nat Commun. 2024 Mar 23;15(1):2626. doi: 10.1038/s41467-024-46917-1. Nat Commun. 2024. PMID: 38521790 Free PMC article.
-
The S-component fold: a link between bacterial transporters and receptors.Commun Biol. 2024 May 21;7(1):610. doi: 10.1038/s42003-024-06295-2. Commun Biol. 2024. PMID: 38773269 Free PMC article.
-
Single-molecule visualization of ATP-induced dynamics of the subunit composition of an ECF transporter complex under turnover conditions.Nat Commun. 2025 May 13;16(1):4448. doi: 10.1038/s41467-025-59674-6. Nat Commun. 2025. PMID: 40360487 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

