CD8-Targeted PET Imaging of Tumor-Infiltrating T Cells in Patients with Cancer: A Phase I First-in-Humans Study of 89Zr-Df-IAB22M2C, a Radiolabeled Anti-CD8 Minibody
- PMID: 34413145
- PMCID: PMC9051598
- DOI: 10.2967/jnumed.121.262485
CD8-Targeted PET Imaging of Tumor-Infiltrating T Cells in Patients with Cancer: A Phase I First-in-Humans Study of 89Zr-Df-IAB22M2C, a Radiolabeled Anti-CD8 Minibody
Abstract
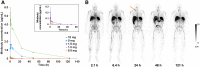
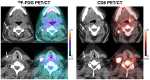
There is a need for in vivo diagnostic imaging probes that can noninvasively measure tumor-infiltrating CD8+ leukocytes. Such imaging probes could be used to predict early response to cancer immunotherapy, help select effective single or combination immunotherapies, and facilitate the development of new immunotherapies or immunotherapy combinations. This study was designed to optimize conditions for performing CD8 PET imaging with 89Zr-Df-IAB22M2C and determine whether CD8 PET imaging could provide a safe and effective noninvasive method of visualizing the whole-body biodistribution of CD8+ leukocytes. Methods: We conducted a phase 1 first-in-humans PET imaging study using an anti-CD8 radiolabeled minibody, 89Zr-Df-IAB22M2C, to detect whole-body and tumor CD8+ leukocyte distribution in patients with metastatic solid tumors. Patients received 111 MBq of 89Zr-Df-IAB22M2C followed by serial PET scanning over 5-7 d. A 2-stage design included a dose-escalation phase and a dose-expansion phase. Biodistribution, radiation dosimetry, and semiquantitative evaluation of 89Zr-Df-IAB22M2C uptake were performed in all patients. Results: Fifteen subjects with metastatic melanoma, non-small cell lung cancer, and hepatocellular carcinoma were enrolled. No drug-related adverse events or abnormal laboratory results were noted except for a transient increase in antidrug antibodies in 1 subject. 89Zr-Df-IAB22M2C accumulated in tumors and CD8-rich tissues (e.g., spleen, bone marrow, nodes), with maximum uptake at 24-48 h after injection and low background activity in CD8-poor tissues (e.g., muscle and lung). Radiotracer uptake in tumors was noted in 10 of 15 subjects, including 7 of 8 subjects on immunotherapy, 1 of 2 subjects on targeted therapy, and 2 of 5 treatment-naïve subjects. In 3 patients with advanced melanoma or hepatocellular carcinoma on immunotherapy, posttreatment CD8 PET/CT scans demonstrated increased 89Zr-Df-IAB22M2C uptake in tumor lesions, which correlated with response. Conclusion: CD8 PET imaging with 89Zr-Df-IAB22M2C is safe and has the potential to visualize the whole-body biodistribution of CD8+ leukocytes in tumors and reference tissues, and may predict early response to immunotherapy.
Trial registration: ClinicalTrials.gov NCT03107663.
Keywords: 89Zr-Df-IAB22M2C; CD8+ T cell; PET imaging; immunotherapy; minibody.
© 2022 by the Society of Nuclear Medicine and Molecular Imaging.
Figures



References
-
- Azimi F, Scolyer RA, Rumcheva P, et al. . Tumor-infiltrating lymphocyte grade is an independent predictor of sentinel lymph node status and survival in patients with cutaneous melanoma. J Clin Oncol. 2012;30:2678–2683. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
