Visuomotor control in mice and primates
- PMID: 34416241
- PMCID: PMC10508359
- DOI: 10.1016/j.neubiorev.2021.08.009
Visuomotor control in mice and primates
Abstract
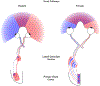
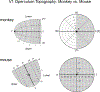
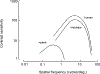
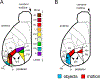
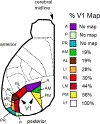
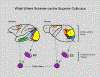
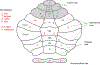
We conduct a comparative evaluation of the visual systems from the retina to the muscles of the mouse and the macaque monkey noting the differences and similarities between these two species. The topics covered include (1) visual-field overlap, (2) visual spatial resolution, (3) V1 cortical point-image [i.e., V1 tissue dedicated to analyzing a unit receptive field], (4) object versus motion encoding, (5) oculomotor range, (6) eye, head, and body movement coordination, and (7) neocortical and cerebellar function. We also discuss blindsight in rodents and primates which provides insights on how the neocortex mediates conscious vision in these species. This review is timely because the field of visuomotor neurophysiology is expanding beyond the macaque monkey to include the mouse; there is therefore a need for a comparative analysis between these two species on how the brain generates visuomotor responses.
Keywords: Blindsight; Cerebellum; Cortical point-image; Macaque monkey; Motion; Mouse; Neocortex; Objects; Oculomotor range.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Figures
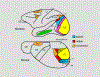
References
-
- Adrian ED, 1922. The relation between the stimulus and the electrical response in a single muscle fiber. Arch. Neérl. Physiol. 7, 330–332.
-
- Adrian ED, 1943. Afferent areas in the cerebellum connected with the limbs. Brain 66, 289–315.
-
- Annese J, Schenker-Ahmed NM, Bartsch H, Maechler P, Sheh C, Thomas N, Kayano J, Ghatan A, Bresler N, Frosch MP, Klaming R, Corkin S, 2014. Postmortem examination of patients H.M.’s brain based on histological sectioning and digital 3D reconstruction. Nature Comm. doi: 10.1038/ncomms4122. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

