Ghrelin protects against lipopolysaccharide-induced acute respiratory distress syndrome through the PI3K/AKT pathway
- PMID: 34437900
- PMCID: PMC8445891
- DOI: 10.1016/j.jbc.2021.101111
Ghrelin protects against lipopolysaccharide-induced acute respiratory distress syndrome through the PI3K/AKT pathway
Abstract
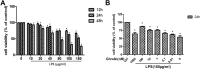
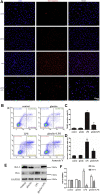
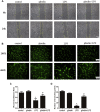
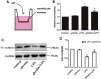
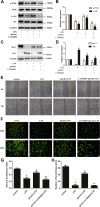
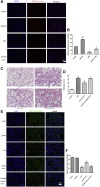
Pulmonary endothelial barrier dysfunction is a major pathophysiology observed in acute respiratory distress syndrome (ARDS). Ghrelin, a key regulator of metabolism, has been shown to play protective roles in the respiratory system. However, its effects on lipopolysaccharide (LPS)-induced pulmonary endothelial barrier injury are unknown. In this study, the effects of ghrelin on LPS-induced ARDS and endothelial cell injury were evaluated in vivo and in vitro. In vivo, mice treated with LPS (3 mg/kg intranasal application) were used to establish the ARDS model. Annexin V/propidium iodide apoptosis assay, scratch-wound assay, tube formation assay, transwell permeability assay, and Western blotting experiment were performed to reveal in vitro effects and underlying mechanisms of ghrelin on endothelial barrier function. Our results showed that ghrelin had protective effects on LPS-induced ARDS and endothelial barrier disruption by inhibiting apoptosis, promoting cell migration and tube formation, and activating the PI3K/AKT signaling pathway. Furthermore, ghrelin stabilized LPS-induced endothelial barrier function by decreasing endothelial permeability and increasing the expression of the intercellular junction protein vascular endothelial cadherin. LY294002, a specific inhibitor of the PI3K pathway, reversed the protective effects of ghrelin on the endothelial cell barrier. In conclusion, our findings indicated that ghrelin protected against LPS-induced ARDS by impairing the pulmonary endothelial barrier partly through activating the PI3K/AKT pathway. Thus, ghrelin may be a valuable therapeutic strategy for the prevention or treatment of ARDS.
Keywords: PI3K; acute respiratory distress syndrome; endothelial cells; ghrelin.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article.
Figures

Similar articles
-
Different concentrations of lipopolysaccharide regulate barrier function through the PI3K/Akt signalling pathway in human pulmonary microvascular endothelial cells.Sci Rep. 2018 Jul 2;8(1):9963. doi: 10.1038/s41598-018-28089-3. Sci Rep. 2018. PMID: 29967433 Free PMC article.
-
Intermedin alleviates the inflammatory response and stabilizes the endothelial barrier in LPS-induced ARDS through the PI3K/Akt/eNOS signaling pathway.Int Immunopharmacol. 2020 Nov;88:106951. doi: 10.1016/j.intimp.2020.106951. Epub 2020 Sep 3. Int Immunopharmacol. 2020. PMID: 32892076
-
Vaspin protects against LPS‑induced ARDS by inhibiting inflammation, apoptosis and reactive oxygen species generation in pulmonary endothelial cells via the Akt/GSK‑3β pathway.Int J Mol Med. 2017 Dec;40(6):1803-1817. doi: 10.3892/ijmm.2017.3176. Epub 2017 Oct 9. Int J Mol Med. 2017. PMID: 29039444 Free PMC article.
-
Experimental and clinical perspectives on glycocalyx integrity and its relation to acute respiratory distress syndrome.Biochim Biophys Acta Mol Basis Dis. 2025 Apr;1871(4):167745. doi: 10.1016/j.bbadis.2025.167745. Epub 2025 Feb 22. Biochim Biophys Acta Mol Basis Dis. 2025. PMID: 39987847 Review.
-
A Razor's Edge: Vascular Responses to Acute Inflammatory Lung Injury/Acute Respiratory Distress Syndrome.Annu Rev Physiol. 2024 Feb 12;86:505-529. doi: 10.1146/annurev-physiol-042222-030731. Annu Rev Physiol. 2024. PMID: 38345908 Free PMC article. Review.
Cited by
-
Rhein-attenuates LPS-induced acute lung injury via targeting NFATc1/Trem2 axis.Inflamm Res. 2023 Jun;72(6):1237-1255. doi: 10.1007/s00011-023-01746-8. Epub 2023 May 22. Inflamm Res. 2023. PMID: 37212865 Free PMC article.
-
Advances in the use of exosomes for the treatment of ALI/ARDS.Front Immunol. 2022 Aug 9;13:971189. doi: 10.3389/fimmu.2022.971189. eCollection 2022. Front Immunol. 2022. PMID: 36016948 Free PMC article. Review.
-
Ghrelin inhibits autophagy mediated by AKT/mTOR pathway to ameliorate retinal angiogenesis induced by high glucose stress.Int J Ophthalmol. 2024 May 18;17(5):785-793. doi: 10.18240/ijo.2024.05.01. eCollection 2024. Int J Ophthalmol. 2024. PMID: 38766333 Free PMC article.
-
Ghrelin system in Alzheimer's disease.Curr Opin Neurobiol. 2023 Feb;78:102655. doi: 10.1016/j.conb.2022.102655. Epub 2022 Dec 15. Curr Opin Neurobiol. 2023. PMID: 36527939 Free PMC article. Review.
-
Overexpression of Wnt5a promoted the protective effect of mesenchymal stem cells on Lipopolysaccharide-induced endothelial cell injury via activating PI3K/AKT signaling pathway.BMC Infect Dis. 2024 Mar 20;24(1):335. doi: 10.1186/s12879-024-09204-4. BMC Infect Dis. 2024. PMID: 38509522 Free PMC article.
References
-
- Yuan S.Y., Rigor R.R. Regulation of endothelial barrier function. Morgan and Claypool Life Sciences; San Rafael, CA: 2011. p. 146. - PubMed
-
- Narula T., Deboisblanc B.P. Ghrelin in critical illness. Am. J. Respir. Cell Mol. Biol. 2015;53:437–442. - PubMed
-
- Liu H., Yu X., Yu S., Kou J. Molecular mechanisms in lipopolysaccharide-induced pulmonary endothelial barrier dysfunction. Int. Immunopharmacol. 2015;29:937–946. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

