Cationic Peptidomimetic Amphiphiles Having a N-Aryl- or N-Naphthyl-1,2,3-Triazole Core Structure Targeting Clostridioides (Clostridium) difficile: Synthesis, Antibacterial Evaluation, and an In Vivo C. difficile Infection Model
- PMID: 34438963
- PMCID: PMC8388771
- DOI: 10.3390/antibiotics10080913
Cationic Peptidomimetic Amphiphiles Having a N-Aryl- or N-Naphthyl-1,2,3-Triazole Core Structure Targeting Clostridioides (Clostridium) difficile: Synthesis, Antibacterial Evaluation, and an In Vivo C. difficile Infection Model
Abstract
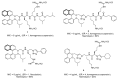
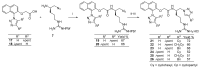
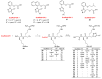
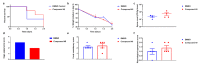
Clostridioides (also known as Clostridium) difficile is a Gram-positive anaerobic, spore producing bacterial pathogen that causes severe gastrointestinal infection in humans. The current chemotherapeutic options are inadequate, expensive, and limited, and thus inexpensive drug treatments for C. difficile infection (CDI) with improved efficacy and specificity are urgently needed. To improve the solubility of our cationic amphiphilic 1,1'-binaphthylpeptidomimetics developed earlier that showed promise in an in vivo murine CDI model we have synthesized related compounds with an N-arytriazole or N-naphthyltriazole moiety instead of the 1,1'-biphenyl or 1,1'-binaphthyl moiety. This modification was made to increase the polarity and thus water solubility of the overall peptidomimetics, while maintaining the aromatic character. The dicationic N-naphthyltriazole derivative 40 was identified as a C. difficile-selective antibacterial with MIC values of 8 µg/mL against C. difficile strains ATCC 700057 and 132 (both ribotype 027). This compound displayed increased water solubility and reduced hemolytic activity (32 µg/mL) in an in vitro hemolysis assay and reduced cytotoxicity (CC50 32 µg/mL against HEK293 cells) relative to lead compound 2. Compound 40 exhibited mild efficacy (with 80% survival observed after 24 h compared to the DMSO control of 40%) in an in vivo murine model of C. difficile infection by reducing the severity and slowing the onset of disease.
Keywords: Clostridioides (Clostridium) difficile; antibacterial; peptidomimetic; triazole.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
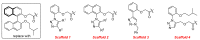
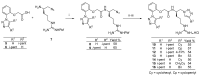
Similar articles
-
Current and Ongoing Developments in Targeting Clostridioides difficile Infection and Recurrence.Microorganisms. 2024 Jun 15;12(6):1206. doi: 10.3390/microorganisms12061206. Microorganisms. 2024. PMID: 38930588 Free PMC article. Review.
-
Cationic biaryl 1,2,3-triazolyl peptidomimetic amphiphiles targeting Clostridioides (Clostridium) difficile: Synthesis, antibacterial evaluation and an in vivo C. difficile infection model.Eur J Med Chem. 2019 May 15;170:203-224. doi: 10.1016/j.ejmech.2019.02.068. Epub 2019 Mar 1. Eur J Med Chem. 2019. PMID: 30901686
-
Binaphthyl-1,2,3-triazole peptidomimetics with activity against Clostridium difficile and other pathogenic bacteria.Org Biomol Chem. 2015 May 28;13(20):5743-56. doi: 10.1039/c5ob00576k. Epub 2015 Apr 22. Org Biomol Chem. 2015. PMID: 25901416
-
Systemic Inflammatory Mediators Are Effective Biomarkers for Predicting Adverse Outcomes in Clostridioides difficile Infection.mBio. 2020 May 5;11(3):e00180-20. doi: 10.1128/mBio.00180-20. mBio. 2020. PMID: 32371595 Free PMC article.
-
Clostridioides (Clostridium) difficile infection: current and alternative therapeutic strategies.Future Microbiol. 2018 Mar;13:469-482. doi: 10.2217/fmb-2017-0203. Epub 2018 Feb 21. Future Microbiol. 2018. PMID: 29464969 Review.
Cited by
-
Current and Ongoing Developments in Targeting Clostridioides difficile Infection and Recurrence.Microorganisms. 2024 Jun 15;12(6):1206. doi: 10.3390/microorganisms12061206. Microorganisms. 2024. PMID: 38930588 Free PMC article. Review.
-
Recent developments in membrane targeting antifungal agents to mitigate antifungal resistance.RSC Med Chem. 2023 Jun 26;14(9):1603-1628. doi: 10.1039/d3md00151b. eCollection 2023 Sep 19. RSC Med Chem. 2023. PMID: 37731690 Free PMC article. Review.
-
Design, Synthesis, and In Vitro Evaluation of the Leishmanicidal Activity of New Aromatic Symmetrical 1,4-Disubstituted 1,2,3-Bistriazoles.ACS Omega. 2025 May 12;10(26):27819-27829. doi: 10.1021/acsomega.5c00492. eCollection 2025 Jul 8. ACS Omega. 2025. PMID: 40657064 Free PMC article.
-
Introduction to the Special Issue on Clostridioides difficile.Antibiotics (Basel). 2021 Oct 11;10(10):1233. doi: 10.3390/antibiotics10101233. Antibiotics (Basel). 2021. PMID: 34680813 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

