Hematopoietic Cell Transplantation for Severe Combined Immunodeficiency Patients: a Japanese Retrospective Study
- PMID: 34448087
- PMCID: PMC8390179
- DOI: 10.1007/s10875-021-01112-5
Hematopoietic Cell Transplantation for Severe Combined Immunodeficiency Patients: a Japanese Retrospective Study
Abstract
Purpose: Hematopoietic cell transplantation (HCT) is a curative therapy for patients with severe combined immunodeficiency (SCID). Here, we conducted a nationwide study to assess the outcome of SCID patients after HCT in Japan.
Methods: A cohort of 181 SCID patients undergoing their first allogeneic HCT in 1974-2016 was studied by using the Japanese national database (Transplant Registry Unified Management Program, TRUMP).
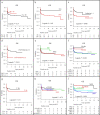
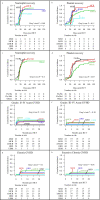
Results: The 10-year overall survival (OS) of the patients who received HCT in 2006-2016 was 67%. Umbilical cord blood (UCB) transplantation was performed in 81 patients (45%). The outcomes of HCT from HLA-matched UCB (n = 21) and matched sibling donors (n = 22) were comparable, including 10-year OS (91% vs. 91%), neutrophil recovery (cumulative incidence at 30 days, 89% vs. 100%), and platelet recovery (cumulative incidence at 60 days, 89% vs. 100%). Multivariate analysis of the patients who received HCT in 2006-2016 demonstrated that the following factors were associated with poor OS: bacterial or fungal infection at HCT (hazard ratio (HR): 3.8, P = 0.006), cytomegalovirus infection prior to HCT (HR: 9.4, P = 0.03), ≥ 4 months of age at HCT (HR: 25.5, P = 0.009), and mismatched UCB (HR: 19.8, P = 0.01).
Conclusion: We showed the potential of HLA-matched UCB as a donor source with higher priority for SCID patients. We also demonstrated that early age at HCT without active infection is critical for a better prognosis, highlighting the importance of newborn screening for SCID.
Keywords: Cord blood transplantation; Hematopoietic cell transplantation; Japan; Newborn screening; Retrospective study; Severe combined immunodeficiency.
© 2021. The Author(s), under exclusive licence to Springer Science+Business Media, LLC, part of Springer Nature.
Conflict of interest statement
The authors declare no competing interest.
Figures
Similar articles
-
Survival After Hematopoietic Stem Cell Transplantation in Severe Combined Immunodeficiency (SCID): A Worldwide Review of the Prognostic Variables.Clin Rev Allergy Immunol. 2024 Apr;66(2):192-209. doi: 10.1007/s12016-024-08993-5. Epub 2024 Apr 30. Clin Rev Allergy Immunol. 2024. PMID: 38689103 Review.
-
Hematopoietic Cell Transplantation for Inborn Errors of Immunity Other than Severe Combined Immunodeficiency in Japan: Retrospective Analysis for 1985-2016.J Clin Immunol. 2022 Apr;42(3):529-545. doi: 10.1007/s10875-021-01199-w. Epub 2022 Jan 4. J Clin Immunol. 2022. PMID: 34981329
-
[Analysis of efficacy and prognosis of allogeneic hematopoietic stem cell transplantation for the treatment of combined immunodeficiency].Zhonghua Er Ke Za Zhi. 2024 May 2;62(5):444-450. doi: 10.3760/cma.j.cn112140-20230815-00105. Zhonghua Er Ke Za Zhi. 2024. PMID: 38623012 Chinese.
-
Single-unit unrelated cord blood transplantation versus HLA-matched sibling transplantation in adults with advanced myelodysplastic syndrome: A registry-based study from the adult MDS working group of the Japanese society for transplantation and cellular therapy.Hematol Oncol. 2024 Jan;42(1):e3217. doi: 10.1002/hon.3217. Epub 2023 Aug 18. Hematol Oncol. 2024. PMID: 37592904
-
Current Knowledge and Priorities for Future Research in Late Effects after Hematopoietic Stem Cell Transplantation (HCT) for Severe Combined Immunodeficiency Patients: A Consensus Statement from the Second Pediatric Blood and Marrow Transplant Consortium International Conference on Late Effects after Pediatric HCT.Biol Blood Marrow Transplant. 2017 Mar;23(3):379-387. doi: 10.1016/j.bbmt.2016.12.619. Epub 2017 Jan 6. Biol Blood Marrow Transplant. 2017. PMID: 28068510 Free PMC article. Review.
Cited by
-
Endocrinopathies in Inborn Errors of Immunity.Front Immunol. 2021 Nov 23;12:786241. doi: 10.3389/fimmu.2021.786241. eCollection 2021. Front Immunol. 2021. PMID: 34887872 Free PMC article. Review.
-
Conditioning regimens for inborn errors of immunity: current perspectives and future strategies.Int J Hematol. 2022 Jul;116(1):7-15. doi: 10.1007/s12185-022-03389-7. Epub 2022 Jun 8. Int J Hematol. 2022. PMID: 35675025 Review.
-
Population pharmacokinetics and initial dose optimization of tacrolimus in children with severe combined immunodeficiency undergoing hematopoietic stem cell transplantation.Front Pharmacol. 2022 Jul 22;13:869939. doi: 10.3389/fphar.2022.869939. eCollection 2022. Front Pharmacol. 2022. PMID: 35935844 Free PMC article.
-
Survival After Hematopoietic Stem Cell Transplantation in Severe Combined Immunodeficiency (SCID): A Worldwide Review of the Prognostic Variables.Clin Rev Allergy Immunol. 2024 Apr;66(2):192-209. doi: 10.1007/s12016-024-08993-5. Epub 2024 Apr 30. Clin Rev Allergy Immunol. 2024. PMID: 38689103 Review.
-
Perspectives in newborn screening for SCID in Japan. Case report: newborn screening identified X-linked severe combined immunodeficiency with a novel IL2RG variant.Front Immunol. 2024 Nov 20;15:1478411. doi: 10.3389/fimmu.2024.1478411. eCollection 2024. Front Immunol. 2024. PMID: 39635533 Free PMC article.
References
-
- Tangye SG, Al-Herz W, Bousfiha A, Chatila T, Cunningham-Rundles C, Etzioni A, et al. Human inborn errors of immunity: 2019 update on the classification from the International Union of Immunological Societies Expert Committee. J Clin Immunol. 2020;40:24–64. doi: 10.1007/s10875-020-00758-x. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials

