Characterization of linear epitope specificity of antibodies potentially contributing to spontaneous clearance of hepatitis C virus
- PMID: 34449828
- PMCID: PMC8396737
- DOI: 10.1371/journal.pone.0256816
Characterization of linear epitope specificity of antibodies potentially contributing to spontaneous clearance of hepatitis C virus
Abstract
Background: Around 30% of the HCV infected patients can spontaneously clear the virus. Cumulative evidence suggests the role of neutralizing antibodies in such spontaneous resolution. Understanding the epitope specificity of such antibodies will inform the rational vaccine design as such information is limited to date. In addition to conformational epitope targeted antibodies, linear epitope specific antibodies have been identified that are broadly cross reactive against diverse HCV strains. In this study, we have characterized the potential role of three conserved linear epitopes in the spontaneous clearance of HCV.
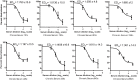
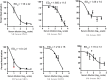
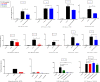
Methods: We tested the reactivity of sera from chronic patients (CP) and spontaneous resolvers (SR) with linear peptides corresponding to three conserved regions of HCV envelope protein E2 spanning amino acids 412-423, 523-532 and 432-443 using ELISA. Subsequently, we characterized the dependency of HCV neutralization by the reactive serum samples on the antibodies specific for these epitopes using pseudoparticle-based neutralization assay. In ELISA most of the CP sera showed reactivity to multiple peptides while most of the SR samples were reactive to a single peptide suggesting presence of more specific antibodies in the SR sera. In most of the HCVpp neutralizing sera of particular peptide reactivity the neutralization was significantly affected by the presence of respective peptide. HCV neutralization by CP sera was affected by multiple peptides while 75% of the HCVpp neutralizing SR sera were competed by the 432 epitope.
Conclusions: These findings suggest that individuals who spontaneously resolve HCV infection at the acute phase, can produce antibodies specific for conserved linear epitopes, and those antibodies can potentially play a role in the spontaneous viral clearance. The epitope present in the 432-443 region of E2 was identified as the primary neutralizing epitope with potential role in spontaneous viral clearance and this epitope potentiates for the design of immunogen for prophylactic vaccine.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- WHO. Global hepatitis report 2017. 2017.
-
- Takehara T, Sakamoto N, Nishiguchi S, Ikeda F, Tatsumi T, Ueno Y, et al.. Efficacy and safety of sofosbuvir-velpatasvir with or without ribavirin in HCV-infected Japanese patients with decompensated cirrhosis: an open-label phase 3 trial. J Gastroenterol. 2019;54(1):87–95. doi: 10.1007/s00535-018-1503-x - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

