A hidden threshold in motor neuron gene networks revealed by modulation of miR-218 dose
- PMID: 34450025
- PMCID: PMC8542606
- DOI: 10.1016/j.neuron.2021.07.028
A hidden threshold in motor neuron gene networks revealed by modulation of miR-218 dose
Abstract
Disruption of homeostatic microRNA (miRNA) expression levels is known to cause human neuropathology. However, the gene regulatory and phenotypic effects of altering a miRNA's in vivo abundance (rather than its binary gain or loss) are not well understood. By genetic combination, we generated an allelic series of mice expressing varying levels of miR-218, a motor neuron-selective gene regulator associated with motor neuron disease. Titration of miR-218 cellular dose unexpectedly revealed complex, non-ratiometric target mRNA dose responses and distinct gene network outputs. A non-linearly responsive regulon exhibited a steep miR-218 dose-dependent threshold in repression that, when crossed, resulted in severe motor neuron synaptic failure and death. This work demonstrates that a miRNA can govern distinct gene network outputs at different expression levels and that miRNA-dependent phenotypes emerge at particular dose ranges because of hidden regulatory inflection points of their underlying gene networks.
Keywords: amyotrophic lateral sclerosis; gene dosage; gene networks; haploinsufficiency; microRNA-218; motoneuron; neurodevelopment; neuromuscular junction; neuropathology; single cell RNA sequencing.
Published by Elsevier Inc.
Conflict of interest statement
Declaration of interests The authors note a related US patent related to motor neuron-specific expression vectors (US Patent 10,729,790).
Figures
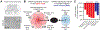




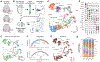

Comment in
-
MiR-218 steps down to a threshold of motor impairment.Neuron. 2021 Oct 20;109(20):3233-3235. doi: 10.1016/j.neuron.2021.09.030. Neuron. 2021. PMID: 34672981
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

