Soft Polymer-Based Technique for Cellular Force Sensing
- PMID: 34451211
- PMCID: PMC8399510
- DOI: 10.3390/polym13162672
Soft Polymer-Based Technique for Cellular Force Sensing
Abstract
Soft polymers have emerged as a vital type of material adopted in biomedical engineering to perform various biomechanical characterisations such as sensing cellular forces. Distinct advantages of these materials used in cellular force sensing include maintaining normal functions of cells, resembling in vivo mechanical characteristics, and adapting to the customised functionality demanded in individual applications. A wide range of techniques has been developed with various designs and fabrication processes for the desired soft polymeric structures, as well as measurement methodologies in sensing cellular forces. This review highlights the merits and demerits of these soft polymer-based techniques for measuring cellular contraction force with emphasis on their quantitativeness and cell-friendliness. Moreover, how the viscoelastic properties of soft polymers influence the force measurement is addressed. More importantly, the future trends and advancements of soft polymer-based techniques, such as new designs and fabrication processes for cellular force sensing, are also addressed in this review.
Keywords: 3D matrix; cell-friendly; cellular biomechanics; force-sensing; hydrogel; soft polymer; tissue engineering.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
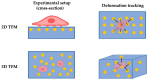
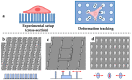





References
-
- Behring J., Junker R., Walboomers X.F., Chessnut B., Jansen J.A. Toward guided tissue and bone regeneration: Morphology, attachment, proliferation, and migration of cells cultured on collagen barrier membranes. A systematic review. Odontology. 2008;96:1–11. doi: 10.1007/s10266-008-0087-y. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

