A Comprehensive Review of Microneedles: Types, Materials, Processes, Characterizations and Applications
- PMID: 34451353
- PMCID: PMC8400269
- DOI: 10.3390/polym13162815
A Comprehensive Review of Microneedles: Types, Materials, Processes, Characterizations and Applications
Abstract
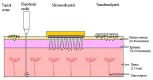
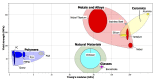
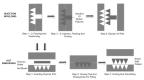
Drug delivery through the skin offers many advantages such as avoidance of hepatic first-pass metabolism, maintenance of steady plasma concentration, safety, and compliance over oral or parenteral pathways. However, the biggest challenge for transdermal delivery is that only a limited number of potent drugs with ideal physicochemical properties can passively diffuse and intercellularly permeate through skin barriers and achieve therapeutic concentration by this route. Significant efforts have been made toward the development of approaches to enhance transdermal permeation of the drugs. Among them, microneedles represent one of the microscale physical enhancement methods that greatly expand the spectrum of drugs for transdermal and intradermal delivery. Microneedles typically measure 0.1-1 mm in length. In this review, microneedle materials, fabrication routes, characterization techniques, and applications for transdermal delivery are discussed. A variety of materials such as silicon, stainless steel, and polymers have been used to fabricate solid, coated, hollow, or dissolvable microneedles. Their implications for transdermal drug delivery have been discussed extensively. However, there remain challenges with sustained delivery, efficacy, cost-effective fabrication, and large-scale manufacturing. This review discusses different modes of characterization and the gaps in manufacturing technologies associated with microneedles. This review also discusses their potential impact on drug delivery, vaccine delivery, disease diagnostic, and cosmetics applications.
Keywords: 3D printing; advanced manufacturing; characterization; drug delivery; microneedle; polymers; therapeutics; transdermal.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures
















References
-
- Ranade V.V., Hollinger M.A., Cannon J.B. Drug Delivery Systems. CRC Press; Boca Raton, FL, USA: 2003.
-
- Hassan B.A.R. Overview on Drug Delivery System. Pharm. Anal. Acta. 2012;3:4172.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

