Innovations and Patent Trends in the Development of USFDA Approved Protein Kinase Inhibitors in the Last Two Decades
- PMID: 34451807
- PMCID: PMC8400070
- DOI: 10.3390/ph14080710
Innovations and Patent Trends in the Development of USFDA Approved Protein Kinase Inhibitors in the Last Two Decades
Abstract
Protein kinase inhibitors (PKIs) are important therapeutic agents. As of 31 May 2021, the United States Food and Drug Administration (USFDA) has approved 70 PKIs. Most of the PKIs are employed to treat cancer and inflammatory diseases. Imatinib was the first PKI approved by USFDA in 2001. This review summarizes the compound patents and the essential polymorph patents of the PKIs approved by the USFDA from 2001 to 31 May 2021. The dates on the generic drug availability of the PKIs in the USA market have also been forecasted. It is expected that 19 and 48 PKIs will be genericized by 2025 and 2030, respectively, due to their compound patent expiry. This may reduce the financial toxicity associated with the existing PKIs. There are nearly 535 reported PKs. However, the USFDA approved PKIs target only about 10-15% of the total said PKs. As a result, there are still a large number of unexplored PKs. As the field advances during the next 20 years, one can anticipate that PKIs with many scaffolds, chemotypes, and pharmacophores will be developed.
Keywords: USFDA; cancer; generic product; inflammation; patent review; protein kinase inhibitors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures












































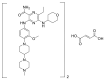















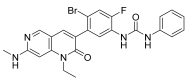
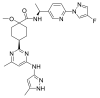

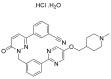




Similar articles
-
Recent Advances in Pyrazole-based Protein Kinase Inhibitors as Emerging Therapeutic Targets.Comb Chem High Throughput Screen. 2024;27(19):2791-2804. doi: 10.2174/0113862073252211231024182817. Comb Chem High Throughput Screen. 2024. PMID: 37946345 Review.
-
PKIDB: A Curated, Annotated and Updated Database of Protein Kinase Inhibitors in Clinical Trials.Molecules. 2018 Apr 15;23(4):908. doi: 10.3390/molecules23040908. Molecules. 2018. PMID: 29662024 Free PMC article.
-
Comparative Assessment of Protein Kinase Inhibitors in Public Databases and in PKIDB.Molecules. 2020 Jul 15;25(14):3226. doi: 10.3390/molecules25143226. Molecules. 2020. PMID: 32679723 Free PMC article.
-
Systematic Analysis of Covalent and Allosteric Protein Kinase Inhibitors.Molecules. 2023 Aug 1;28(15):5805. doi: 10.3390/molecules28155805. Molecules. 2023. PMID: 37570774 Free PMC article.
-
Assessment of acute kidney injury related to small-molecule protein kinase inhibitors using the FDA adverse event reporting system.Cancer Chemother Pharmacol. 2020 Nov;86(5):655-662. doi: 10.1007/s00280-020-04151-8. Epub 2020 Oct 1. Cancer Chemother Pharmacol. 2020. PMID: 33001273
Cited by
-
Discovery, Development, Inventions and Patent Review of Fexinidazole: The First All-Oral Therapy for Human African Trypanosomiasis.Pharmaceuticals (Basel). 2022 Jan 21;15(2):128. doi: 10.3390/ph15020128. Pharmaceuticals (Basel). 2022. PMID: 35215241 Free PMC article. Review.
-
Oral Brincidofovir Therapy for Monkeypox Outbreak: A Focused Review on the Therapeutic Potential, Clinical Studies, Patent Literature, and Prospects.Biomedicines. 2023 Jan 19;11(2):278. doi: 10.3390/biomedicines11020278. Biomedicines. 2023. PMID: 36830816 Free PMC article. Review.
-
Highlights on the Development, Related Patents, and Prospects of Lenacapavir: The First-in-Class HIV-1 Capsid Inhibitor for the Treatment of Multi-Drug-Resistant HIV-1 Infection.Medicina (Kaunas). 2023 May 28;59(6):1041. doi: 10.3390/medicina59061041. Medicina (Kaunas). 2023. PMID: 37374245 Free PMC article. Review.
-
Targeting Protein Kinases and Epigenetic Control as Combinatorial Therapy Options for Advanced Prostate Cancer Treatment.Pharmaceutics. 2022 Feb 25;14(3):515. doi: 10.3390/pharmaceutics14030515. Pharmaceutics. 2022. PMID: 35335890 Free PMC article. Review.
-
Natural Products in Precision Oncology: Plant-Based Small Molecule Inhibitors of Protein Kinases for Cancer Chemoprevention.Nutrients. 2023 Feb 27;15(5):1192. doi: 10.3390/nu15051192. Nutrients. 2023. PMID: 36904191 Free PMC article. Review.
References
-
- Keohane E.M., Otto C.N., Walenga J.M. Rodak’s Hematology-E-Book: Clinical Principles and Applications. Elsevier Health Sciences; Amsterdam, The Netherlands: 2019.
-
- Prescott J.C., Braisted A. Identification of Kinase Inhibitors. Application Publication Number WO2005034840A2. PCT Patent. 2005 Apr 21;
-
- Charrier J.D., Durrant S. Protein Kinase Inhibitors. Application Publication Number US20120028966A1. U.S. Patent. 2012 Feb 2;
-
- Plowman G., Whyte D., Manning G., Sudarsanam S., Martinez R. Novel human protein kinases and protein kinase-like enzymes. Application Publication Number US20040048310A1. U.S. Patent. 2004 Mar 11;
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

