Synthesis and Biological Activities of Pyrazino[1,2- a]indole and Pyrazino[1,2- a]indol-1-one Derivatives
- PMID: 34451876
- PMCID: PMC8399128
- DOI: 10.3390/ph14080779
Synthesis and Biological Activities of Pyrazino[1,2- a]indole and Pyrazino[1,2- a]indol-1-one Derivatives
Abstract
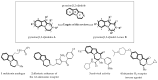
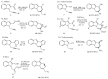
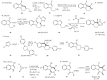
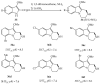
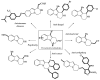
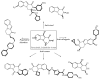
This review concerns the synthesis and biological activities of pyrazino[1,2-a]indoles and pyrazino[1,2-a]indol-1-ones reported since 1997 and the discovery of biological activity of pyrazinoindole derivatives. In the first part, we first presented the synthetic routes that have been reported from a methodological point of view to access the pyrazinoindole unit according to cyclization reactions using or not using metal catalysts. Then, syntheses and neuropsychiatric, auto-immune, anti-infectious and anti-cancer properties of pyrazinoindoles were detailed. In the second part, we first reported the main accesses to pyrazinoindol-1-one substrates according to Michael reactions, metal-catalyzed and metal-free cyclization reactions. The syntheses and anti-cancer, anti-infectious, anti-allergenic and neuropsychiatric properties of pyrazinoindolones were next described and discussed.
Keywords: biological activity; catalysis; cyclization; pyrazinoindole; pyrazinoindolone.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Intramolecular cyclization of delta-iminoacetylenes: a new entry to pyrazino[1,2-a]indoles.J Org Chem. 2005 May 13;70(10):4088-95. doi: 10.1021/jo0502246. J Org Chem. 2005. PMID: 15876101
-
Chiral Pd-Catalyzed Enantioselective Syntheses of Various N-C Axially Chiral Compounds and Their Synthetic Applications.Acc Chem Res. 2021 Feb 2;54(3):719-730. doi: 10.1021/acs.accounts.0c00767. Epub 2021 Jan 22. Acc Chem Res. 2021. PMID: 33481580
-
Base-Mediated, Chemo- and Regioselective (4+2) Annulation of Indole-2-carboxamides with 2,3-Epoxy Tosylates toward 1,2-Fused Indoles.J Org Chem. 2023 Jul 7;88(13):9237-9248. doi: 10.1021/acs.joc.3c00813. Epub 2023 Jun 8. J Org Chem. 2023. PMID: 37289967
-
Indole Derivatives as Anti-Tubercular Agents: An Overview on their Synthesis and Biological Activities.Curr Med Chem. 2021;28(22):4531-4568. doi: 10.2174/0929867327666200918144709. Curr Med Chem. 2021. PMID: 32951569 Review.
-
Solid phase synthesis of biologically important indoles.Curr Med Chem. 2009;16(20):2531-65. doi: 10.2174/092986709788682010. Curr Med Chem. 2009. PMID: 19601797 Review.
Cited by
-
The Current Landscape in the Development of Small-molecule Modulators Targeting Sphingosine-1-phosphate Receptors to Treat Neurodegenerative Diseases.Curr Top Med Chem. 2024;24(28):2431-2446. doi: 10.2174/0115680266288509240422112839. Curr Top Med Chem. 2024. PMID: 38676503 Review.
-
Highly regioselective and diastereoselective synthesis of novel pyrazinoindolones via a base-mediated Ugi-N-alkylation sequence.RSC Adv. 2023 Jun 6;13(25):16963-16969. doi: 10.1039/d3ra02065g. eCollection 2023 Jun 5. RSC Adv. 2023. PMID: 37288378 Free PMC article.
-
Synthesis of 3-substituted 2,3-dihydropyrazino[1,2-a]indol-4(1H)-ones by sequential reactions of 2-indolylmethyl acetates with α-amino acids.RSC Adv. 2023 Mar 29;13(15):10090-10096. doi: 10.1039/d3ra01335a. eCollection 2023 Mar 27. RSC Adv. 2023. PMID: 37006346 Free PMC article.
-
New Potential Agents for Malignant Melanoma Treatment-Most Recent Studies 2020-2022.Int J Mol Sci. 2022 May 29;23(11):6084. doi: 10.3390/ijms23116084. Int J Mol Sci. 2022. PMID: 35682764 Free PMC article. Review.
References
-
- Singh A., Mahapatra S., Sewariya S., Singh N., Singh S., Kumar Y., Bandichhor R., Chandra R. A mini-review on the synthesis of pyrazinoindole: Recent progress and perspectives. Mini Rev. Org. Chem. 2021;18:504–514. doi: 10.2174/1570193X17999200806151209. - DOI
-
- Sokolova E.A., Festa A.A. Synthesis of pyrazino[1,2-a] indoles and indolo [1,2-a] quinoxalines (microreview) Chem. Heterocycl. Comp. 2016;52:219–221. doi: 10.1007/s10593-016-1866-y. - DOI
-
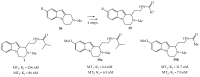
- Markl C., Attia M.I., Julius J., Sehti S., Witt-Enderby P.A., Zlotos D.P. Synthesis and pharmacological evaluation of 1,2,3,4-tetrahydropyrazino[1,2-a] indole and 2-[(phenylmethylamino)methyl]-1H-indole analogues as novel melatoninergic ligands. Bioorg. Med. Chem. 2009;17:4583–4594. doi: 10.1016/j.bmc.2009.04.068. - DOI - PubMed
-
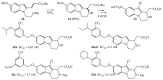
- Romagnoli R., Baraldi P.G., Carrion M.D., Cara C.L., Salvador M.K., Preti D., Tabrizi M.A., Moorman A.R., Vincenzi F., Borea P.A., et al. Synthesis and biological effects of novel 2-amino-3-(4-chlorobenzoyl)-4-substituted thiophenes as allosteric enhancers of the A1 adenosine receptor. Eur. J. Med. Chem. 2013;67:409–427. doi: 10.1016/j.ejmech.2013.07.002. - DOI - PubMed
-
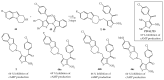
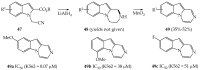
- Kounde C.S., Yeo H.Q., Wang Q.Y., Wan K.F., Dong H., Karuna R., Dix I., Wagner T., Zou B., Simon O. Discovery of 2-oxopiperazine dengue inhibitors by scaffold morphing of a phenotypic high-throughput screening hit. Bioorg. Med. Chem. Lett. 2017;27:1385–1389. doi: 10.1016/j.bmcl.2017.02.005. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

