Experimental and Theoretical Study on Theobromine Solubility Enhancement in Binary Aqueous Solutions and Ternary Designed Solvents
- PMID: 34452079
- PMCID: PMC8401494
- DOI: 10.3390/pharmaceutics13081118
Experimental and Theoretical Study on Theobromine Solubility Enhancement in Binary Aqueous Solutions and Ternary Designed Solvents
Abstract
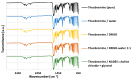
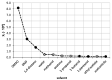
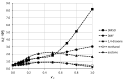
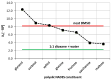
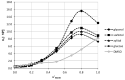
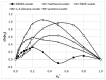
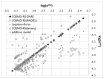
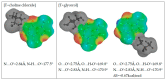
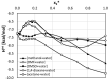
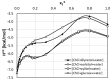
The solubility of theobromine was studied both experimentally and theoretically. The solubility was determined spectrophotometrically at 25 °C in neat organic solvents, aqueous binary mixtures, Natural Deep Eutectic Solvents (NADES) and ternary NADES mixtures with water. It was found that addition of water in unimolar proportions with some organic solvents increases theobromine solubility compared to neat solvents. Additionally, using NADES results in a solubility increase of the studied compound not only in relation to water but also DMSO. The addition of water (0.2 molar fraction) to NADES is responsible for an even larger increase of solubility. The measured solubilities were interpreted in terms of three theoretical frameworks. The first one-belonging to the set of data reduction techniques-proved to be very efficient in quantitative back-computations of excess solubility of theobromine in all studied systems. The default approach utilizing the well-recognized COSMO-RS (Conductor-like Screening Model for Real Solvents) framework offered at most a qualitative solubility description. The extended search for possible contacts provided evidence for the existence of many intermolecular complexes that alter the electron density of the solute molecule, thus influencing solubility computations. Taking into account such intermolecular contacts by using the COSMO-RS-DARE (Conductor-like Screening Model for Realistic Solvation-Dimerization, Aggregation, and Reaction Extension) framework seriously increased the accuracy of solubility computations.
Keywords: COSMO-RS; NADES; methylxanthines; solubility; theobromine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Solvent Screening for Solubility Enhancement of Theophylline in Neat, Binary and Ternary NADES Solvents: New Measurements and Ensemble Machine Learning.Int J Mol Sci. 2021 Jul 8;22(14):7347. doi: 10.3390/ijms22147347. Int J Mol Sci. 2021. PMID: 34298966 Free PMC article.
-
Thermodynamics and Intermolecular Interactions of Nicotinamide in Neat and Binary Solutions: Experimental Measurements and COSMO-RS Concentration Dependent Reactions Investigations.Int J Mol Sci. 2021 Jul 8;22(14):7365. doi: 10.3390/ijms22147365. Int J Mol Sci. 2021. PMID: 34298985 Free PMC article.
-
Solubility advantage of sulfanilamide and sulfacetamide in natural deep eutectic systems: experimental and theoretical investigations.Drug Dev Ind Pharm. 2019 Jul;45(7):1120-1129. doi: 10.1080/03639045.2019.1597104. Epub 2019 Apr 2. Drug Dev Ind Pharm. 2019. PMID: 30883240
-
Solubility prediction, solvate and cocrystal screening as tools for rational crystal engineering.J Pharm Pharmacol. 2015 Jun;67(6):803-11. doi: 10.1111/jphp.12376. Epub 2015 Apr 7. J Pharm Pharmacol. 2015. PMID: 25851032 Review.
-
Modeling the Physicochemical Properties of Natural Deep Eutectic Solvents.ChemSusChem. 2020 Aug 7;13(15):3789-3804. doi: 10.1002/cssc.202000286. Epub 2020 Jun 22. ChemSusChem. 2020. PMID: 32378359 Review.
Cited by
-
Application of the Solute-Solvent Intermolecular Interactions as Indicator of Caffeine Solubility in Aqueous Binary Aprotic and Proton Acceptor Solvents: Measurements and Quantum Chemistry Computations.Materials (Basel). 2022 Mar 27;15(7):2472. doi: 10.3390/ma15072472. Materials (Basel). 2022. PMID: 35407805 Free PMC article.
-
Experimental and Machine-Learning-Assisted Design of Pharmaceutically Acceptable Deep Eutectic Solvents for the Solubility Improvement of Non-Selective COX Inhibitors Ibuprofen and Ketoprofen.Molecules. 2024 May 14;29(10):2296. doi: 10.3390/molecules29102296. Molecules. 2024. PMID: 38792157 Free PMC article.
-
Intermolecular Interactions of Edaravone in Aqueous Solutions of Ethaline and Glyceline Inferred from Experiments and Quantum Chemistry Computations.Molecules. 2023 Jan 7;28(2):629. doi: 10.3390/molecules28020629. Molecules. 2023. PMID: 36677688 Free PMC article.
-
Exploration of the Solubility Hyperspace of Selected Active Pharmaceutical Ingredients in Choline- and Betaine-Based Deep Eutectic Solvents: Machine Learning Modeling and Experimental Validation.Molecules. 2024 Oct 16;29(20):4894. doi: 10.3390/molecules29204894. Molecules. 2024. PMID: 39459262 Free PMC article.
-
Experimental and Theoretical Screening for Green Solvents Improving Sulfamethizole Solubility.Materials (Basel). 2021 Oct 9;14(20):5915. doi: 10.3390/ma14205915. Materials (Basel). 2021. PMID: 34683507 Free PMC article.
References
-
- Andreeva E.Y., Dmitrienko S.G., Zolotov Y.A. Methylxanthines: Properties and determination in various objects. Russ. Chem. Rev. 2012;81:397–414. doi: 10.1070/RC2012v081n05ABEH004220. - DOI
-
- Spiller G.A. Caffeine. 1st ed. CRC Press; Boca Raton, FL, USA: 1998.
-
- Craig C.R., Stitzel R.E. Modern Pharmacology with Clinical Applications. 6th ed. Lippincott Williams and Wilkins; Philadelphia, PA, USA: 2003.
-
- Satoskar R.S., Rege N., Bhandarkar S.D. Pharmacology and Pharmacotherapeutics. 24th ed. Elsevier; New Delhi, India: 2015.
LinkOut - more resources
Full Text Sources

