Exploring the Multifunctional Roles of Odontoglossum Ringspot Virus P126 in Facilitating Cymbidium Mosaic Virus Cell-to-Cell Movement during Mixed Infection
- PMID: 34452417
- PMCID: PMC8402721
- DOI: 10.3390/v13081552
Exploring the Multifunctional Roles of Odontoglossum Ringspot Virus P126 in Facilitating Cymbidium Mosaic Virus Cell-to-Cell Movement during Mixed Infection
Abstract
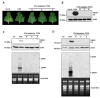
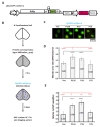
Synergistic interactions among viruses, hosts and/or transmission vectors during mixed infection can alter viral titers, symptom severity or host range. Viral suppressors of RNA silencing (VSRs) are considered one of such factors contributing to synergistic responses. Odontoglossum ringspot virus (ORSV) and cymbidium mosaic virus (CymMV), which are two of the most significant orchid viruses, exhibit synergistic symptom intensification in Phalaenopsis orchids with unilaterally enhanced CymMV movement by ORSV. In order to reveal the underlying mechanisms, we generated infectious cDNA clones of ORSV and CymMV isolated from Phalaenopsis that exerted similar unilateral synergism in both Phalaenopsis orchid and Nicotiana benthamiana. Moreover, we show that the ORSV replicase P126 is a VSR. Mutagenesis analysis revealed that mutation of the methionine in the carboxyl terminus of ORSV P126 abolished ORSV replication even though some P126 mutants preserved VSR activity, indicating that the VSR function of P126 alone is not sufficient for viral replication. Thus, P126 functions in both ORSV replication and as a VSR. Furthermore, P126 expression enhanced cell-to-cell movement and viral titers of CymMV in infected Phalaenopsis flowers and N. benthamiana leaves. Taking together, both the VSR and protein function of P126 might be prerequisites for unilaterally enhancing CymMV cell-to-cell movement by ORSV.
Keywords: Phalaenopsis; RNA silencing; cymbidium mosaic virus (CymMV); odontoglossum ringspot virus (ORSV); viral suppressor of RNA silencing (VSR); viral synergism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Genome-wide analysis of small RNAs from Odontoglossum ringspot virus and Cymbidium mosaic virus synergistically infecting Phalaenopsis.Mol Plant Pathol. 2020 Feb;21(2):188-205. doi: 10.1111/mpp.12888. Epub 2019 Nov 14. Mol Plant Pathol. 2020. PMID: 31724809 Free PMC article.
-
Dual resistance of transgenic plants against Cymbidium mosaic virus and Odontoglossum ringspot virus.Sci Rep. 2019 Jul 15;9(1):10230. doi: 10.1038/s41598-019-46695-7. Sci Rep. 2019. PMID: 31308424 Free PMC article.
-
Reciprocal function of movement proteins and complementation of long-distance movement of Cymbidium mosaic virus RNA by Odontoglossum ringspot virus coat protein.J Gen Virol. 2005 May;86(Pt 5):1543-1553. doi: 10.1099/vir.0.80772-0. J Gen Virol. 2005. PMID: 15831968
-
Virus resistance in orchids.Plant Sci. 2014 Nov;228:26-38. doi: 10.1016/j.plantsci.2014.04.015. Epub 2014 Apr 28. Plant Sci. 2014. PMID: 25438783 Review.
-
Viral and nonviral elements in potexvirus replication and movement and in antiviral responses.Adv Virus Res. 2013;87:75-112. doi: 10.1016/B978-0-12-407698-3.00003-X. Adv Virus Res. 2013. PMID: 23809921 Review.
Cited by
-
Special Issue "State-of-the-Art Plant-Virus Interactions in Asia".Viruses. 2022 Apr 21;14(5):864. doi: 10.3390/v14050864. Viruses. 2022. PMID: 35632605 Free PMC article.
-
Role of Plant Virus Movement Proteins in Suppression of Host RNAi Defense.Int J Mol Sci. 2023 May 20;24(10):9049. doi: 10.3390/ijms24109049. Int J Mol Sci. 2023. PMID: 37240394 Free PMC article. Review.
References
-
- Vance V.B. Replication of Potato virus-X RNA is altered in coinfections with Potato virus-Y. Virology. 1991;182:486–494. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources

