Precision dosing of intravenous busulfan in pediatric hematopoietic stem cell transplantation: Results from a multicenter population pharmacokinetic study
- PMID: 34453497
- PMCID: PMC8452291
- DOI: 10.1002/psp4.12683
Precision dosing of intravenous busulfan in pediatric hematopoietic stem cell transplantation: Results from a multicenter population pharmacokinetic study
Abstract
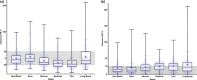
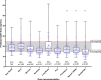
Busulfan (Bu) is a common component of conditioning regimens before hematopoietic stem cell transplantation (HSCT) and is known for high interpatient pharmacokinetic (PK) variability. This study aimed to develop and externally validate a multicentric, population PK (PopPK) model for intravenous Bu in pediatric patients before HSCT to first study the influence of glutathione-s-transferase A1 (GSTA1) polymorphisms on Bu's PK in a large multicentric pediatric population while accounting for fludarabine (Flu) coadministration and, second, to establish an individualized, model-based, first-dose recommendation for intravenous Bu that can be widely used in pediatric patients. The model was built using data from 302 patients from five transplantation centers who received a Bu-based conditioning regimen. External model validation used data from 100 patients. The relationship between body weight and Bu clearance (CL) was best described by an age-dependent allometric scaling of a body weight model. A stepwise covariate analysis identified Day 1 of Bu conditioning, GSTA1 metabolic groups based on GSTA1 polymorphisms, and Flu coadministration as significant covariates influencing Bu CL. The final model adequately predicted Bu first-dose CL in the external cohort, with 81% of predicted area under the curves within the therapeutic window. The final model showed minimal bias (mean prediction error, -0.5%; 95% confidence interval [CI], -3.1% to 2.0%) and acceptable precision (mean absolute prediction error percentage, 18.7%; 95% CI, 17.0%-20.5%) in Bu CL prediction for dosing. This multicentric PopPK study confirmed the influence of GSTA1 polymorphisms and Flu coadministration on Bu CL. The developed model accurately predicted Bu CL and first doses in an external cohort of pediatric patients.
© 2021 The Authors. CPT: Pharmacometrics & Systems Pharmacology published by Wiley Periodicals LLC on behalf of American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
The authors declared no competing interests for this work. Dr. Nastya Kassir was employed by Certara in the past. She is now employed by Genentech/Roche.
Figures


Similar articles
-
Incorporation of GSTA1 genetic variations into a population pharmacokinetic model for IV busulfan in paediatric hematopoietic stem cell transplantation.Br J Clin Pharmacol. 2018 Jul;84(7):1494-1504. doi: 10.1111/bcp.13566. Epub 2018 Apr 27. Br J Clin Pharmacol. 2018. PMID: 29469189 Free PMC article.
-
Pharmacokinetic behavior and appraisal of intravenous busulfan dosing in infants and older children: the results of a population pharmacokinetic study from a large pediatric cohort undergoing hematopoietic stem-cell transplantation.Ther Drug Monit. 2012 Apr;34(2):198-208. doi: 10.1097/FTD.0b013e31824c2f60. Ther Drug Monit. 2012. PMID: 22406655
-
Population pharmacokinetic analysis of intravenous busulfan: GSTA1 genotype is not a predictive factor of initial dose in Chinese adult patients undergoing hematopoietic stem cell transplantation.Cancer Chemother Pharmacol. 2020 Feb;85(2):293-308. doi: 10.1007/s00280-019-04001-2. Epub 2019 Dec 13. Cancer Chemother Pharmacol. 2020. PMID: 31834435
-
Therapeutic drug monitoring of busulfan in transplantation.Curr Pharm Des. 2008;14(20):1936-49. doi: 10.2174/138161208785061382. Curr Pharm Des. 2008. PMID: 18691105 Review.
-
Pharmacogenetic aspects of drug metabolizing enzymes in busulfan based conditioning prior to allogenic hematopoietic stem cell transplantation in children.Curr Drug Metab. 2014 Mar;15(3):251-64. doi: 10.2174/1389200215666140202214012. Curr Drug Metab. 2014. PMID: 24524663 Review.
Cited by
-
Population Pharmacokinetics of Busulfan and Its Metabolite Sulfolane in Patients with Myelofibrosis Undergoing Hematopoietic Stem Cell Transplantation.Pharmaceutics. 2022 May 27;14(6):1145. doi: 10.3390/pharmaceutics14061145. Pharmaceutics. 2022. PMID: 35745718 Free PMC article.
-
Total Body Irradiation Forever? Optimising Chemotherapeutic Options for Irradiation-Free Conditioning for Paediatric Acute Lymphoblastic Leukaemia.Front Pediatr. 2021 Dec 10;9:775485. doi: 10.3389/fped.2021.775485. eCollection 2021. Front Pediatr. 2021. PMID: 34956984 Free PMC article. Review.
-
Optimal Once-Daily Busulfan Administration in Pediatric Patients: A Simulation-Based Investigation of Intravenous Infusion Times.Drug Des Devel Ther. 2024 Mar 20;18:871-879. doi: 10.2147/DDDT.S451970. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 38524879 Free PMC article.
-
Utilization of a Population Pharmacokinetic Model to Improve a Busulfan Test-Dose Strategy in Allogeneic Hematopoietic Cell Transplant Recipients.J Clin Pharmacol. 2023 Sep;63(9):1026-1035. doi: 10.1002/jcph.2257. Epub 2023 Jun 9. J Clin Pharmacol. 2023. PMID: 37122163 Free PMC article.
-
Population Pharmacokinetic Modeling for Twice-Daily Intravenous Busulfan in a Large Cohort of Pediatric Patients Undergoing Hematopoietic Stem Cell Transplantation-A 10-Year Single-Center Experience.Pharmaceutics. 2023 Dec 20;16(1):0. doi: 10.3390/pharmaceutics16010013. Pharmaceutics. 2023. PMID: 38276491 Free PMC article.
References
-
- Slattery JT, Sanders JE, Buckner CD, et al. Graft‐rejection and toxicity following bone marrow transplantation in relation to busulfan pharmacokinetics. Bone Marrow Transplant. 1995;16:31‐42. - PubMed
-
- Slattery JT, Risler LJ. Therapeutic monitoring of busulfan in hematopoietic stem cell transplantation. Ther Drug Monit. 1998;20:543‐549. - PubMed
-
- Dix SP, Wingard JR, Mullins RE, et al. Association of busulfan area under the curve with veno‐occlusive disease following BMT. Bone Marrow Transplant. 1996;17:225‐230. - PubMed
-
- McCune JS, Gooley T, Gibbs JP, et al. Busulfan concentration and graft rejection in pediatric patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant. 2002;30:167‐173. - PubMed
-
- Bolinger AM, Zangwill AB, Slattery JT, et al. Target dose adjustment of busulfan in pediatric patients undergoing bone marrow transplantation. Bone Marrow Transplant. 2001;28:1013‐1018. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

