High-level resistance to bictegravir and cabotegravir in subtype A- and D-infected HIV-1 patients failing raltegravir with multiple resistance mutations
- PMID: 34453542
- PMCID: PMC8521396
- DOI: 10.1093/jac/dkab276
High-level resistance to bictegravir and cabotegravir in subtype A- and D-infected HIV-1 patients failing raltegravir with multiple resistance mutations
Abstract
Objectives: The second-generation integrase strand transfer inhibitor (INSTI) bictegravir is becoming accessible in low- and middle-income countries (LMICs), and another INSTI, cabotegravir, has recently been approved as a long-acting injectable. Data on bictegravir and cabotegravir susceptibility in raltegravir-experienced HIV-1 subtype A- and D-infected patients carrying drug resistance mutations (DRMs) remain very scarce in LMICs.
Patients and methods: HIV-1 integrase (IN)-recombinant viruses from eight patients failing raltegravir-based third-line therapy in Uganda were genotypically and phenotypically tested for susceptibility to bictegravir and cabotegravir. Ability of these viruses to integrate into human genomes was assessed in MT-4 cells.
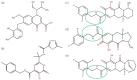
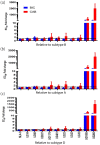
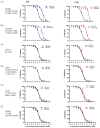
Results: HIV-1 IN-recombinant viruses harbouring single primary mutations (N155H or Y143R/S) or in combination with secondary INSTI mutations (T97A, M50I, L74IM, E157Q, G163R or V151I) were susceptible to both bictegravir and cabotegravir. However, combinations of primary INSTI-resistance mutations such as E138A/G140A/G163R/Q148R or E138K/G140A/S147G/Q148K led to decreased susceptibility to both cabotegravir (fold change in EC50 values from 429 to 1000×) and bictegravir (60 to 100×), exhibiting a high degree of cross-resistance. However, these same IN-recombinant viruses showed impaired integration capacity (14% to 48%) relative to the WT HIV-1 NL4-3 strain in the absence of drug.
Conclusions: Though not currently widely accessible in most LMICs, bictegravir and cabotegravir offer a valid alternative to HIV-infected individuals harbouring subtype A and D HIV-1 variants with reduced susceptibility to first-generation INSTIs but previous exposure to raltegravir may reduce efficacy, more so with cabotegravir.
© The Author(s) 2021. Published by Oxford University Press on behalf of the British Society for Antimicrobial Chemotherapy. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures


References
-
- WHO. Update of recommendations on first- and second-line antiretroviral regimens. 2019. https://apps.who.int/iris/bitstream/handle/10665/325892/WHO-CDS-HIV-19.1....
-
- Gilead. U.S. Food and Drug Administration Approves Gilead’s Biktarvy® (Bictegravir, Emtricitabine, Tenofovir Alafenamide) for Treatment of HIV-1 Infection. 2018. https://www.gilead.com/news-and-press/press-room/press-releases/2018/2/u....
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

