Evaluation of the Cepheid Xpert C. difficile diagnostic assay: an update meta-analysis
- PMID: 34455573
- PMCID: PMC8578272
- DOI: 10.1007/s42770-021-00563-7
Evaluation of the Cepheid Xpert C. difficile diagnostic assay: an update meta-analysis
Abstract
Background: Accurate and rapid diagnosis of Clostridium difficile infection (CDI) is critical for effective patient management and implementation of infection control measures to prevent transmission.
Objectives: We updated our previous meta-analysis to provide a more reliable evidence base for the clinical diagnosis of Xpert C. difficile (Xpert C. difficile) assay.
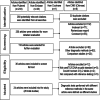
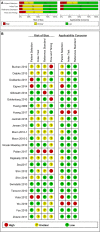
Methods: We searched PubMed, EMBASE, Cochrane Library, Chinese National Knowledge Infrastructure (CNKI), and the Chinese Biomedical Literature Database (CBM) databases to identify studies according to predetermined criteria. STATA 13.0 software was used to analyze the tests for sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and area under the summary receiver operating characteristic curves (AUC). QUADAS-2 was used to assess the quality of included studies with RevMan 5.2. Heterogeneity in accuracy measures was tested with Spearman correlation coefficient and chi-square. Meta-regressions and subgroup analyses were performed to figure out the potential sources of heterogeneity. Model diagnostics were used to evaluate the veracity of the data.
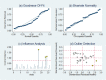
Results: A total of 26 studies were included in the meta-analysis. The pooled sensitivity (95% confidence intervals [CI]) for diagnosis was 0.97(0.95-0.98), and specificity was 0.96(0.95-0.97). The AUC was 0.99 (0.98-1.00). Model diagnostics confirmed the robustness of our meta-analysis's results. Significant heterogeneity was still observed when we pooled most of the accuracy measures of selected studies. Meta-regression and subgroup analyses showed that the sample size and type, ethnicity, and disease prevalence might be the conspicuous sources of heterogeneity.
Conclusions: The up-to-date meta-analysis showed the Xpert CD assay had good accuracy for detecting CDI. However, the diagnosis of CDI must combine clinical presentation with diagnostic testing to better answer the question of whether the patient actually has CDI in the future, and inclusion of preanalytical parameters and clinical outcomes in study design would provide a more objective evidence base.
Keywords: Clostridium infections; Meta-analysis; Nucleic acid amplification techniques.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Accuracy of Xpert Clostridium difficile assay for the diagnosis of Clostridium difficile infection: A meta analysis.PLoS One. 2017 Oct 9;12(10):e0185891. doi: 10.1371/journal.pone.0185891. eCollection 2017. PLoS One. 2017. PMID: 29016644 Free PMC article.
-
Diagnostic accuracy of loop-mediated isothermal amplification in detection of Clostridium difficile in stool samples: a meta-analysis.Arch Med Sci. 2015 Oct 12;11(5):927-36. doi: 10.5114/aoms.2015.54846. Arch Med Sci. 2015. PMID: 26528332 Free PMC article. Review.
-
The Clinical and Laboratory Impact of Upgrading Clostridioides (formerly Clostridium) difficile Infection Testing from Routine to Molecular Based-Algorithm: an Observational Case-Study from the Eastern Province, Saudi Arabia.Clin Lab. 2019 Aug 1;65(8). doi: 10.7754/Clin.Lab.2019.181252. Clin Lab. 2019. PMID: 31414736
-
Diagnostic accuracy of real-time polymerase chain reaction in detection of Clostridium difficile in the stool samples of patients with suspected Clostridium difficile Infection: a meta-analysis.Clin Infect Dis. 2011 Oct;53(7):e81-90. doi: 10.1093/cid/cir505. Clin Infect Dis. 2011. PMID: 21890762
-
Pooled Analysis of the Accuracy of Xpert Ebola Assay for Diagnosing Ebola Virus Infection.Biomed Res Int. 2021 May 17;2021:5527505. doi: 10.1155/2021/5527505. eCollection 2021. Biomed Res Int. 2021. PMID: 34055977 Free PMC article. Review.
Cited by
-
Analysis of high-molecular-weight proteins using MALDI-TOF MS and machine learning for the differentiation of clinically relevant Clostridioides difficile ribotypes.Eur J Clin Microbiol Infect Dis. 2025 Feb;44(2):417-425. doi: 10.1007/s10096-024-05023-2. Epub 2024 Dec 17. Eur J Clin Microbiol Infect Dis. 2025. PMID: 39688756
-
Automated web-based typing of Clostridioides difficile ribotypes via MALDI-TOF MS.BMC Bioinformatics. 2025 Jul 17;26(1):181. doi: 10.1186/s12859-025-06200-6. BMC Bioinformatics. 2025. PMID: 40676564 Free PMC article.
References
-
- Miller BA, Chen LF, Sexton DJ, Anderson DJ. Comparison of the burdens of hospital-onset, healthcare facility-associated Clostridium difficile infection and of healthcare-associated infection due to methicillin-resistant Staphylococcus aureus in community hospitals. Infect Control Hosp Epidemiol. 2011;32:387–390. - PubMed
-
- Barrett ML, Owens PL (2018) Clostridium difficile hospitalizations, 2011–2015. US Agency for Healthcare Research and Quality
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

