Harnessing cyclotides to design and develop novel peptide GPCR ligands
- PMID: 34458757
- PMCID: PMC8341132
- DOI: 10.1039/d0cb00062k
Harnessing cyclotides to design and develop novel peptide GPCR ligands
Abstract
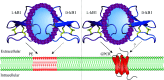
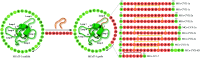
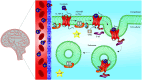
Cyclotides are plant-derived cyclic, disulfide-rich peptides with a unique cyclic cystine knot topology that confers them with remarkable structural stability and resistance to proteolytic degradation. Recently, cyclotides have emerged as promising scaffold molecules for designing peptide-based therapeutics. Here, we provide examples of how engineering cyclotides using molecular grafting may lead to the development of novel peptide ligands of G protein-coupled receptors (GPCRs), today's most exploited drug targets. Integrating bioactive epitopes into stable cyclotide scaffolds can lead to improved pharmacokinetics and oral activity as well as selectivity and high enzymatic stability. We also discuss and highlight the importance of engineered cyclotides as novel tools to study GPCR signaling.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures






References
-
- Gupta A. Gomes I. Bobeck E. N. Fakira A. K. Massaro N. P. Sharma I. Cave A. Hamm H. E. Parello J. Devi L. A. Collybolide is a novel biased agonist of kappa-opioid receptors with potent antipruritic activity. Proc. Natl. Acad. Sci. U. S. A. 2016;113:6041. doi: 10.1073/pnas.1521825113. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

