Artificial intelligence for MRI diagnosis of joints: a scoping review of the current state-of-the-art of deep learning-based approaches
- PMID: 34467424
- PMCID: PMC8692303
- DOI: 10.1007/s00256-021-03830-8
Artificial intelligence for MRI diagnosis of joints: a scoping review of the current state-of-the-art of deep learning-based approaches
Abstract
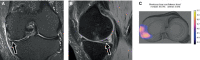
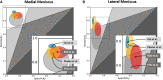
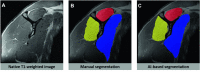
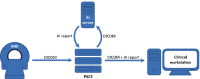
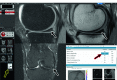
Deep learning-based MRI diagnosis of internal joint derangement is an emerging field of artificial intelligence, which offers many exciting possibilities for musculoskeletal radiology. A variety of investigational deep learning algorithms have been developed to detect anterior cruciate ligament tears, meniscus tears, and rotator cuff disorders. Additional deep learning-based MRI algorithms have been investigated to detect Achilles tendon tears, recurrence prediction of musculoskeletal neoplasms, and complex segmentation of nerves, bones, and muscles. Proof-of-concept studies suggest that deep learning algorithms may achieve similar diagnostic performances when compared to human readers in meta-analyses; however, musculoskeletal radiologists outperformed most deep learning algorithms in studies including a direct comparison. Earlier investigations and developments of deep learning algorithms focused on the binary classification of the presence or absence of an abnormality, whereas more advanced deep learning algorithms start to include features for characterization and severity grading. While many studies have focused on comparing deep learning algorithms against human readers, there is a paucity of data on the performance differences of radiologists interpreting musculoskeletal MRI studies without and with artificial intelligence support. Similarly, studies demonstrating the generalizability and clinical applicability of deep learning algorithms using realistic clinical settings with workflow-integrated deep learning algorithms are sparse. Contingent upon future studies showing the clinical utility of deep learning algorithms, artificial intelligence may eventually translate into clinical practice to assist detection and characterization of various conditions on musculoskeletal MRI exams.
Keywords: Artificial intelligence; Computer; Deep learning; Joints; Magnetic resonance imaging; Musculoskeletal system; Neural networks.
© 2021. The Author(s).
Conflict of interest statement
Benjamin Fritz: none. Jan Fritz: Jan Fritz received institutional research support from Siemens AG, BTG International, Zimmer Biomed, DePuy Synthes, QED, and SyntheticMR; is a scientific advisor for Siemens AG, SyntheticMR, GE Healthcare, QED, BTG, ImageBiopsy Lab, Boston Scientific, and Mirata Pharma; and has shared patents with Siemens Healthcare and Johns Hopkins University.
Figures

References
-
- Gore JC. Artificial intelligence in medical imaging. Magn Reson Imaging. 2020;68:A1–A4. - PubMed
-
- Lee CS, Nagy PG, Weaver SJ, Newman-Toker DE. Cognitive and system factors contributing to diagnostic errors in radiology. AJR Am J Roentgenol. 2013;201(3):611–617. - PubMed
-
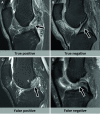
- Germann C, Marbach G, Civardi F, Fucentese SF, Fritz J, Sutter R, et al. Deep convolutional neural network-based diagnosis of anterior cruciate ligament tears: performance comparison of homogenous versus heterogeneous knee MRI cohorts with different pulse sequence protocols and 1.5-T and 3-T magnetic field strengths. Invest Radiol. 2020; 55(8):499–506. - PMC - PubMed
-
- Fritz J, Germann, C., Sutter R, Fritz B. AI-augmented MRI diagnosis of ACL tears: which readers benefit? SSR Annual Meeting 2021.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

