Pharmacological Tuning of Adenosine Signal Nuances Underlying Heart Failure With Preserved Ejection Fraction
- PMID: 34489711
- PMCID: PMC8417789
- DOI: 10.3389/fphar.2021.724320
Pharmacological Tuning of Adenosine Signal Nuances Underlying Heart Failure With Preserved Ejection Fraction
Abstract
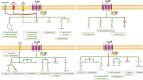
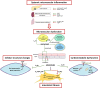
Heart failure with preserved ejection fraction (HFpEF) roughly represents half of the cardiac failure events in developed countries. The proposed 'systemic microvascular paradigm' has been used to explain HFpHF presentation heterogeneity. The lack of effective treatments with few evidence-based therapeutic recommendations makes HFpEF one of the greatest unmet clinical necessities worldwide. The endogenous levels of the purine nucleoside, adenosine, increase significantly following cardiovascular events. Adenosine exerts cardioprotective, neuromodulatory, and immunosuppressive effects by activating plasma membrane-bound P1 receptors that are widely expressed in the cardiovascular system. Its proven benefits have been demonstrated in preclinical animal tests. Here, we provide a comprehensive and up-to-date critical review about the main therapeutic advantages of tuning adenosine signalling pathways in HFpEF, without discounting their side effects and how these can be seized.
Keywords: adenosine; adenosine receptor; cardiac comorbidities; cardiac fibrosis and hypertrophy; endothelial dysfunction; preserved ejection fraction heart failure.
Copyright © 2021 Campos-Martins, Bragança, Correia-de-Sá and Fontes-Sousa.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation.J Am Coll Cardiol. 2013 Jul 23;62(4):263-71. doi: 10.1016/j.jacc.2013.02.092. Epub 2013 May 15. J Am Coll Cardiol. 2013. PMID: 23684677 Review.
-
Cardiomyocyte Functional Etiology in Heart Failure With Preserved Ejection Fraction Is Distinctive-A New Preclinical Model.J Am Heart Assoc. 2018 Jun 1;7(11):e007451. doi: 10.1161/JAHA.117.007451. J Am Heart Assoc. 2018. PMID: 29858360 Free PMC article.
-
Endothelial Dysfunction in Heart Failure With Preserved Ejection Fraction: What are the Experimental Proofs?Front Physiol. 2022 Jul 8;13:906272. doi: 10.3389/fphys.2022.906272. eCollection 2022. Front Physiol. 2022. PMID: 35874523 Free PMC article. Review.
-
Heart failure with preserved ejection fraction: controversies, challenges and future directions.Heart. 2018 Mar;104(5):377-384. doi: 10.1136/heartjnl-2016-310790. Epub 2018 Jan 5. Heart. 2018. PMID: 29305560 Review.
-
Rationale and design of the phase 2b clinical trials to study the effects of the partial adenosine A1-receptor agonist neladenoson bialanate in patients with chronic heart failure with reduced (PANTHEON) and preserved (PANACHE) ejection fraction.Eur J Heart Fail. 2018 Nov;20(11):1601-1610. doi: 10.1002/ejhf.1295. Epub 2018 Sep 17. Eur J Heart Fail. 2018. PMID: 30225882 Clinical Trial.
Cited by
-
Adenosine A3 Receptor: From Molecular Signaling to Therapeutic Strategies for Heart Diseases.Int J Mol Sci. 2024 May 25;25(11):5763. doi: 10.3390/ijms25115763. Int J Mol Sci. 2024. PMID: 38891948 Free PMC article. Review.
-
International Union of Basic and Clinical Pharmacology. CXII: Adenosine Receptors: A Further Update.Pharmacol Rev. 2022 Apr;74(2):340-372. doi: 10.1124/pharmrev.121.000445. Pharmacol Rev. 2022. PMID: 35302044 Free PMC article. Review.
References
-
- Ahmad A., Schaack J. B., White C. W., Ahmad S. (2013). Adenosine A2A Receptor-dependent Proliferation of Pulmonary Endothelial Cells Is Mediated through Calcium Mobilization, PI3-Kinase and ERK1/2 Pathways. Biochem. Biophys. Res. Commun. 434 (3), 566–571. 10.1016/j.bbrc.2013.03.115 - DOI - PMC - PubMed
-
- Alencar A. K., Carvalho F. I., Silva A. M., Martinez S. T., Calasans-Maia J. A., Fraga C. M., et al. (2018). Synergistic Interaction between a PDE5 Inhibitor (Sildenafil) and a New Adenosine A2A Receptor Agonist (LASSBio-1359) Improves Pulmonary Hypertension in Rats. PLoS One 13 (4), e0195047. 10.1371/journal.pone.0195047 - DOI - PMC - PubMed
-
- Alencar A. K. N., Pereira S. L., da Silva F. E., Mendes L. V. P., Cunha V. d. M. N., Lima L. M., et al. (2014). N-acylhydrazone Derivative Ameliorates Monocrotaline-Induced Pulmonary Hypertension through the Modulation of Adenosine AA2R Activity. Int. J. Cardiol. 173 (2), 154–162. 10.1016/j.ijcard.2014.02.022 - DOI - PubMed
-
- Alencar A. K. N., Pereira S. L., Montagnoli T. L., Maia R. C., Kümmerle A. E., Landgraf S. S., et al. (2013). Beneficial Effects of a Novel Agonist of the Adenosine A2Areceptor on Monocrotaline-Induced Pulmonary Hypertension in Rats. Br. J. Pharmacol. 169 (5), 953–962. 10.1111/bph.12193 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

