Biocatalytic Asymmetric Cyclopropanations via Enzyme-Bound Iminium Ion Intermediates
- PMID: 34490955
- PMCID: PMC8596749
- DOI: 10.1002/anie.202110719
Biocatalytic Asymmetric Cyclopropanations via Enzyme-Bound Iminium Ion Intermediates
Abstract
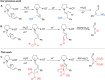
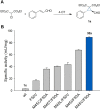
Cyclopropane rings are an important structural motif frequently found in many natural products and pharmaceuticals. Commonly, biocatalytic methodologies for the asymmetric synthesis of cyclopropanes rely on repurposed or artificial heme enzymes. Here, we engineered an unusual cofactor-independent cyclopropanation enzyme based on a promiscuous tautomerase for the enantioselective synthesis of various cyclopropanes via the nucleophilic addition of diethyl 2-chloromalonate to α,β-unsaturated aldehydes. The engineered enzyme promotes formation of the two new carbon-carbon bonds with excellent stereocontrol over both stereocenters, affording the desired cyclopropanes with high diastereo- and enantiopurity (d.r. up to 25:1; e.r. up to 99:1). Our results highlight the usefulness of promiscuous enzymes for expanding the biocatalytic repertoire for non-natural reactions.
Keywords: biocatalysis; catalytic promiscuity; cyclopropanation; enzyme engineering.
© 2021 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

