Elucidating the Role of Cerebellar Synaptic Dysfunction in C9orf72-ALS/FTD - a Systematic Review and Meta-Analysis
- PMID: 34491551
- PMCID: PMC9325807
- DOI: 10.1007/s12311-021-01320-0
Elucidating the Role of Cerebellar Synaptic Dysfunction in C9orf72-ALS/FTD - a Systematic Review and Meta-Analysis
Abstract
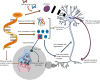
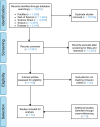
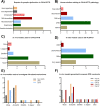
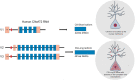
A hexanucleotide repeat expansion in the C9orf72 gene is the most common genetic cause of amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD) with synaptic dysfunction identified as an early pathological hallmark. Although TDP-43 pathology and overt neurodegeneration are largely absent from the cerebellum, the pathological hallmarks of RNA foci and dipeptide repeat protein (DPR) inclusions are most abundant. Here, we present a systematic literature search in the databases of PubMed, Scopus, Embase, Web of Science and Science Direct up until March 5, 2021, which yielded 19,515 publications. Following the exclusion criteria, 72 articles were included having referred to C9orf72, synapses and the cerebellum. Meta-analyses were conducted on studies which reported experimental and control groups with means and standard deviations extracted from figures using the online tool PlotDigitizer. This revealed dendritic defects (P = 0.03), reduced C9orf72 in human patients (P = 0.005) and DPR-related neuronal loss (P = 0.0006) but no neuromuscular junction abnormalities (P = 0.29) or cerebellar neuronal loss (P = 0.23). Our results suggest that dendritic arborisation defects, synaptic gene dysregulation and altered synaptic neurotransmission may drive cerebellar synaptic dysfunction in C9-ALS/FTD. In this review, we discuss how the chronological appearance of the different pathological hallmarks alters synaptic integrity which may have profound implications for disease progression. We conclude that a reduction in C9orf72 protein levels combined with the accumulation of RNA foci and DPRs act synergistically to drive C9 synaptopathy in the cerebellum of C9-ALS/FTD patients.
Keywords: ALS; C9orf72; Cerebellum; FTD; Synaptic dysfunction.
© 2021. Crown.
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
A robust evaluation of TDP-43, poly GP, cellular pathology and behavior in an AAV-C9ORF72 (G4C2)66 mouse model.Acta Neuropathol Commun. 2024 Dec 26;12(1):203. doi: 10.1186/s40478-024-01911-y. Acta Neuropathol Commun. 2024. PMID: 39722074 Free PMC article.
-
Prevalence of brain and spinal cord inclusions, including dipeptide repeat proteins, in patients with the C9ORF72 hexanucleotide repeat expansion: a systematic neuropathological review.Neuropathol Appl Neurobiol. 2016 Oct;42(6):547-60. doi: 10.1111/nan.12284. Neuropathol Appl Neurobiol. 2016. PMID: 26373655
-
Repeated mild traumatic brain injury triggers pathology in asymptomatic C9ORF72 transgenic mice.Brain. 2023 Dec 1;146(12):5139-5152. doi: 10.1093/brain/awad264. Brain. 2023. PMID: 37527465 Free PMC article.
-
Molecular pathology, developmental changes and synaptic dysfunction in (pre-) symptomatic human C9ORF72-ALS/FTD cerebral organoids.Acta Neuropathol Commun. 2024 Sep 18;12(1):152. doi: 10.1186/s40478-024-01857-1. Acta Neuropathol Commun. 2024. PMID: 39289761 Free PMC article.
-
C9orf72 Frontotemporal Dementia and/or Amyotrophic Lateral Sclerosis.2015 Jan 8 [updated 2020 Dec 17]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2015 Jan 8 [updated 2020 Dec 17]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 25577942 Free Books & Documents. Review.
Cited by
-
Cerebellum in Alzheimer's disease and other neurodegenerative diseases: an emerging research frontier.MedComm (2020). 2024 Jul 13;5(7):e638. doi: 10.1002/mco2.638. eCollection 2024 Jul. MedComm (2020). 2024. PMID: 39006764 Free PMC article. Review.
-
Pathology-specific patterns of cerebellar atrophy in neurodegenerative disorders.Alzheimers Dement. 2024 Mar;20(3):1771-1783. doi: 10.1002/alz.13551. Epub 2023 Dec 18. Alzheimers Dement. 2024. PMID: 38109286 Free PMC article.
-
Hyperactivity of Purkinje cell and motor deficits in C9orf72 knockout mice.Mol Cell Neurosci. 2022 Jul;121:103756. doi: 10.1016/j.mcn.2022.103756. Epub 2022 Jul 16. Mol Cell Neurosci. 2022. PMID: 35843530 Free PMC article.
-
Synaptopathy: presynaptic convergence in frontotemporal dementia and amyotrophic lateral sclerosis.Brain. 2024 Jul 5;147(7):2289-2307. doi: 10.1093/brain/awae074. Brain. 2024. PMID: 38451707 Free PMC article. Review.
-
Cognitive and behavioral involvement in ALS has been known for more than a century.Neurol Sci. 2022 Dec;43(12):6741-6760. doi: 10.1007/s10072-022-06340-0. Epub 2022 Sep 2. Neurol Sci. 2022. PMID: 36053340 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

