Treatment patterns in people with cystic fibrosis: have they changed since the introduction of ivacaftor?
- PMID: 34497037
- PMCID: PMC9097695
- DOI: 10.1016/j.jcf.2021.08.014
Treatment patterns in people with cystic fibrosis: have they changed since the introduction of ivacaftor?
Abstract
Background: In late 2012, ivacaftor became available in the UK for people with cystic fibrosis (CF) aged 6 years and over with a G551D mutation. Long-term changes in treatment patterns have not previously been reported. We investigated long-term treatment patterns in people with CF with a G551D mutation who took ivacaftor and compared these with non-ivacaftor-treated cohorts using the UK Cystic Fibrosis Registry.
Methods: Using 2007-2018 data we compared treatment patterns between four cohorts: 1: ivacaftor-treated; 2: ivacaftor era (2013-2018), ineligible genotype (no G551D mutation); 3: pre-ivacaftor era (2007-2012), eligible genotype (G551D mutation); 4: pre-ivacaftor era, ineligible genotype. Treatments included: inhaled antibiotics, dornase alfa, hypertonic saline, chronic oral antibiotics and supplementary feeding.
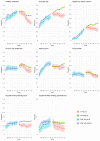
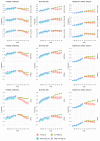
Results: Up to 2012 the percentages of people taking each treatment were similar between the two cohorts defined by genotype and tended to increase by year with a similar slope. Once ivacaftor was introduced, the use of other treatments tended to decrease or remain stable by year for the ivacaftor-treated cohort, whereas it remained stable or increased in the non-ivacaftor-treated cohort. This led to differences in treatment use between the two cohorts in the ivacaftor-era, which became more marked over time.
Conclusions: We have shown a clear divergence in treatment patterns since the introduction of ivacaftor in a number of key treatments widely used in CF. Further research is needed to investigate whether the differences in treatment patterns are associated with changes in health outcomes.
Keywords: cystic fibrosis; ivacaftor; registry data; treatment burden; treatment patterns.
Copyright © 2021. Published by Elsevier B.V.
Conflict of interest statement
Conflict of interest statement GD has received personal fees from Chiesi Limited for lectures, unrelated to the current work. She is co-Chief Investigator for the CF STORM clinical trial. EG has no conflicts of interest to declare. RHK received funding from a Circle of Care Award from Vertex.
Figures
References
-
- Davies J., Sheridan H., Bell N., Cunningham S., Davis S.D., Elborn J.S. Assessment of clinical response to ivacaftor with lung clearance index in cystic fibrosis patients with a G551D-CFTR mutation and preserved spirometry: a randomised controlled trial. Lancet Respir Med. 2013;1(8):630–638. - PubMed
-
- Sawicki G.S., McKone E.F., Pasta D.J., Millar S.J., Wagener J.S., Johnson C.A. Sustained Benefit from ivacaftor demonstrated by combining clinical trial and cystic fibrosis patient registry data. Am J Respir Crit Care Med. 2015;192(7):836–842. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

