Alterations of Ultra Long-Chain Fatty Acids in Hereditary Skin Diseases-Review Article
- PMID: 34497816
- PMCID: PMC8420999
- DOI: 10.3389/fmed.2021.730855
Alterations of Ultra Long-Chain Fatty Acids in Hereditary Skin Diseases-Review Article
Abstract
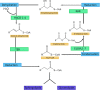
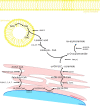
The skin is a flexible organ that forms a barrier between the environment and the body's interior; it is involved in the immune response, in protection and regulation, and is a dynamic environment in which skin lipids play an important role in maintaining homeostasis. The different layers of the skin differ in both the composition and amount of lipids. The epidermis displays the best characteristics in this respect. The main lipids in this layer are cholesterol, fatty acids (FAs) and ceramides. FAs can occur in free form and as components of complex molecules. The most poorly characterized FAs are very long-chain fatty acids (VLCFAs) and ultra long-chain fatty acids (ULCFAs). VLCFAs and ULCFAs are among the main components of ceramides and are part of the free fatty acid (FFA) fraction. They are most abundant in the brain, liver, kidneys, and skin. VLCFAs and ULCFAs are responsible for the rigidity and impermeability of membranes, forming the mechanically and chemically strong outer layer of cell membranes. Any changes in the composition and length of the carbon chains of FAs result in a change in their melting point and therefore a change in membrane permeability. One of the factors causing a decrease in the amount of VLCFAs and ULCFAs is an improper diet. Another much more important factor is mutations in the genes which code proteins involved in the metabolism of VLCFAs and ULCFAs-regarding their elongation, their attachment to ceramides and their transformation. These mutations have their clinical consequences in the form of inborn errors in metabolism and neurodegenerative disorders, among others. Some of them are accompanied by skin symptoms such as ichthyosis and ichthyosiform erythroderma. In the following review, the structure of the skin is briefly characterized and the most important lipid components of the skin are presented. The focus is also on providing an overview of selected proteins involved in the metabolism of VLCFAs and ULCFAs in the skin.
Keywords: ceramides; cholesterol; dermis; epidermis; fatty acids; lipids; skin.
Copyright © 2021 Zwara, Wertheim-Tysarowska and Mika.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
The ELOVL proteins: Very and ultra long-chain fatty acids at the crossroads between metabolic and neurodegenerative disorders.Mol Genet Metab. 2025 Mar;144(3):109050. doi: 10.1016/j.ymgme.2025.109050. Epub 2025 Feb 4. Mol Genet Metab. 2025. PMID: 39946831 Review.
-
The role of very long chain fatty acids in yeast physiology and human diseases.Biol Chem. 2020 Nov 23;402(1):25-38. doi: 10.1515/hsz-2020-0234. Print 2020 Nov 18. Biol Chem. 2020. PMID: 33544487 Review.
-
Conformations of Three Types of Ultra-Long-Chain Fatty Acids in Multicomponent Lipid Bilayers.J Phys Chem B. 2022 Nov 17;126(45):9316-9324. doi: 10.1021/acs.jpcb.2c06189. Epub 2022 Nov 5. J Phys Chem B. 2022. PMID: 36334092
-
Very long-chain fatty acids: elongation, physiology and related disorders.J Biochem. 2012 Nov;152(5):387-95. doi: 10.1093/jb/mvs105. Epub 2012 Sep 14. J Biochem. 2012. PMID: 22984005 Review.
-
Very Long Chain Lipids Favor the Formation of a Homogeneous Phase in Stratum Corneum Model Membranes.Langmuir. 2020 Nov 24;36(46):13899-13907. doi: 10.1021/acs.langmuir.0c02305. Epub 2020 Nov 10. Langmuir. 2020. PMID: 33170015
Cited by
-
Changes in the Serum Fatty Acid Profile After Anhepatic Phase of Orthotopic Liver Transplantation Procedure.Front Physiol. 2022 Mar 29;13:817987. doi: 10.3389/fphys.2022.817987. eCollection 2022. Front Physiol. 2022. PMID: 35422709 Free PMC article.
-
CTIP2 and lipid metabolism: regulation in skin development and associated diseases.Expert Rev Proteomics. 2021 Nov;18(11):1009-1017. doi: 10.1080/14789450.2021.2003707. Epub 2021 Nov 17. Expert Rev Proteomics. 2021. PMID: 34739354 Free PMC article. Review.
-
Comprehensive Molecular Analysis of Disease-Related Genes as First-Tier Test for Early Diagnosis, Classification, and Management of Patients Affected by Nonsyndromic Ichthyosis.Biomedicines. 2024 May 17;12(5):1112. doi: 10.3390/biomedicines12051112. Biomedicines. 2024. PMID: 38791074 Free PMC article.
-
Advanced In Vitro Three-Dimensional Skin Models of Atopic Dermatitis.Tissue Eng Regen Med. 2023 Jul;20(4):539-552. doi: 10.1007/s13770-023-00532-1. Epub 2023 Mar 30. Tissue Eng Regen Med. 2023. PMID: 36995643 Free PMC article. Review.
-
Acetyl-CoA synthesis in the skin is a key determinant of systemic lipid homeostasis.Cell Rep. 2025 Feb 25;44(2):115284. doi: 10.1016/j.celrep.2025.115284. Epub 2025 Feb 10. Cell Rep. 2025. PMID: 39932848 Free PMC article.
References
-
- Biga LM, Dawson S, Hardwell A, Hopkins R, Kaufmann J, LeMaster M, et al. . Anatomy & Physiology. Corvallis, OR: (2015).
-
- Kanitakis J. Anatomy, histology and immunohistochemistry of normal human skin. Eur J Dermatol. (2002) 12:390–9; quiz 400–1. Available online at: https://www.jle.com/en/revues/ejd/e-docs/anatomy_histology_and_immunohis... - PubMed
-
- Barbieri JS, Wanat K, Seykora J. Skin: basic structure and function. In: McManus LM, Mitchell RN. editors. Pathobiology of Human Disease. Amsterdam: Elsevier; (2014). p. 1134–44. 10.1016/B978-0-12-386456-7.03501-2 - DOI
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

