Anakinra versus Baricitinib: Different Strategies for Patients Hospitalized with COVID-19
- PMID: 34501467
- PMCID: PMC8432474
- DOI: 10.3390/jcm10174019
Anakinra versus Baricitinib: Different Strategies for Patients Hospitalized with COVID-19
Abstract
Background: Immunomodulatory drugs have been used in patients with severe COVID-19. The objective of this study was to evaluate the effects of two different strategies, based either on an interleukin-1 inhibitor, anakinra, or on a JAK inhibitor, such as baricitinib, on the survival of patients hospitalized with COVID-19 pneumonia.
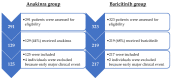
Methods: Individuals admitted to two hospitals because of COVID-19 were included if they fulfilled the clinical, radiological, and laboratory criteria for moderate-to-severe disease. Patients were classified according to the first immunomodulatory drug prescribed: anakinra or baricitinib. All subjects were concomitantly treated with corticosteroids, in addition to standard care. The main outcomes were the need for invasive mechanical ventilation (IMV) and in-hospital death. Statistical analysis included propensity score matching and Cox regression model.
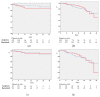
Results: The study subjects included 125 and 217 individuals in the anakinra and baricitinib groups, respectively. IMV was required in 13 (10.4%) and 10 (4.6%) patients, respectively (p = 0.039). During this period, 22 (17.6%) and 36 (16.6%) individuals died in both groups (p = 0.811). Older age, low functional status, high comorbidity, need for IMV, elevated lactate dehydrogenase, and use of a high flow of oxygen at initially were found to be associated with worse clinical outcomes. No differences according to the immunomodulatory therapy used were observed. For most of the deceased individuals, early interruption of anakinra or baricitinib had occurred at the time of their admission to the intensive care unit.
Conclusions: Similar mortality is observed in patients treated with anakinra or baricitinib plus corticosteroids.
Keywords: COVID-19; anakinra; baricitinib; corticosteroids; mortality.
Conflict of interest statement
The authors declare no conflict of interest.
Figures