Hypofractionated Radiotherapy Upregulates Several Immune Checkpoint Molecules in Head and Neck Squamous Cell Carcinoma Cells Independently of the HPV Status While ICOS-L Is Upregulated Only on HPV-Positive Cells
- PMID: 34502022
- PMCID: PMC8430967
- DOI: 10.3390/ijms22179114
Hypofractionated Radiotherapy Upregulates Several Immune Checkpoint Molecules in Head and Neck Squamous Cell Carcinoma Cells Independently of the HPV Status While ICOS-L Is Upregulated Only on HPV-Positive Cells
Abstract
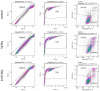
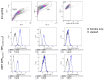
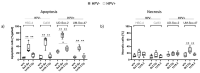
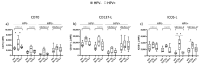
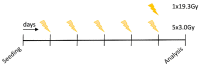
While the treatment of squamous cell carcinoma of the head and neck (HNSCC) with radiotherapy (RT) is complemented more and more by immunotherapy in clinical trials, little is known about the impact of the human papillomavirus (HPV) status or the applied RT scheme on the immune phenotype of the tumor cells. Therefore, we aimed to examine the impact of the HPV status of four human HNSCC cell lines on cell death and the expression of immune checkpoint molecules (ICMs) after RT with either hypofractionation irradiation (5x3.0Gy) or a high single dose (1x19.3Gy) via multicolor flow cytometry and quantitative PCR at an early time point after therapy. In our study, 5x3.0Gy RT induced high numbers of early and late apoptotic cells independent of the HPV status, but necrosis was only increased in the HPV-positive UM-Scc-47 cells. Generally, the immune stimulatory ICMs (CD70, CD137-L, ICOS-L) were less affected by RT compared to the immune suppressive ones (PD-L1, PD-L2, and the herpesvirus entry mediator (HVEM)). A significant higher surface expression of the analyzed ICMs was found after hypofractionated RT compared to a single high dose; however, regardless of the HPV status, with the exception of ICOS-L. Here, HPV-positive HNSCC tumor cells showed a stronger response to 5x3.0Gy than HPV-negative ones. On the RNA level, only minor alterations of ICMs were observed following RT, with the exception of the HPV negative cell line CAL33 treated with 5x3.0Gy, where PD-L2, HVEM and CD70 were significantly increased. We conclude that the HPV status may not distinctly predict immunological responses following RT, and thus cannot be used as a single predictive marker for therapy responses in HNSCC. In contrast, the patient-specific individual expression of ICMs following RT is preferable for the targeted patient selection for immune therapy directed against distinct ICM.
Keywords: HNSCC; HPV status; immune checkpoint molecules; immunotherapy; radiotherapy.
Conflict of interest statement
M.H. (Michael Hader), U.S.G. and R.F. received support for presentation activities from -Ing. Sennewald Medizintechnik GmbH. R.F., U.S.G. and B.F. have received support for investigator initiated clinical studies (IITs) from Merck Sharp & Dohme and AstraZeneca. R.F. and U.S.G. also contributed at Advisory Boards Meetings of AstraZeneca and Bristol-Myers Squibb. M.H. (Markus Hecht): Merck Serono (advisory role, speakers’ bureau, honoraria, travel expenses, research funding); MSD (advisory role, speakers’ bureau, honoraria, travel expenses, research funding); AstraZeneca (research funding); Novartis (research funding); BMS (advisory role, honoraria, speakers’ bureau); Teva (travel expenses). S.W., L.D., A.D., F.G., A.-O.G. and T.W. have no conflict of interest.
Figures


Similar articles
-
Radiotherapy combined with docetaxel alters the immune phenotype of HNSCC cells and results in increased surface expression of CD137 and release of HMGB1 of specifically HPV-positive tumor cells.Neoplasia. 2023 Nov;45:100944. doi: 10.1016/j.neo.2023.100944. Epub 2023 Oct 17. Neoplasia. 2023. PMID: 37857049 Free PMC article.
-
Immune checkpoint expression on tumor-infiltrating lymphocytes (TIL) is dependent on HPV status in oropharyngeal carcinoma (OPSCC) - A single-cell RNA sequencing analysis.Oral Oncol. 2024 Dec;159:107107. doi: 10.1016/j.oraloncology.2024.107107. Epub 2024 Nov 15. Oral Oncol. 2024. PMID: 39549431
-
TLR9 Mediated Tumor-Stroma Interactions in Human Papilloma Virus (HPV)-Positive Head and Neck Squamous Cell Carcinoma Up-Regulate PD-L1 and PD-L2.Front Immunol. 2019 Jul 16;10:1644. doi: 10.3389/fimmu.2019.01644. eCollection 2019. Front Immunol. 2019. PMID: 31379843 Free PMC article.
-
Programmed Death-1/Programmed Death-Ligand 1-Axis Blockade in Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma Stratified by Human Papillomavirus Status: A Systematic Review and Meta-Analysis.Front Immunol. 2021 Apr 7;12:645170. doi: 10.3389/fimmu.2021.645170. eCollection 2021. Front Immunol. 2021. PMID: 33897693 Free PMC article.
-
The radiobiology of HPV-positive and HPV-negative head and neck squamous cell carcinoma.Expert Rev Mol Med. 2020 Jul 2;22:e3. doi: 10.1017/erm.2020.4. Expert Rev Mol Med. 2020. PMID: 32611474 Free PMC article. Review.
Cited by
-
To Die or Not to Die: Cell Death in Biology and Disease.Int J Mol Sci. 2022 Jun 16;23(12):6734. doi: 10.3390/ijms23126734. Int J Mol Sci. 2022. PMID: 35743170 Free PMC article.
-
Modulation of PD‑L1 expression by standard therapy in head and neck cancer cell lines and exosomes.Int J Oncol. 2023 Sep;63(3):102. doi: 10.3892/ijo.2023.5550. Epub 2023 Jul 28. Int J Oncol. 2023. PMID: 37503786 Free PMC article.
-
Defining intra-tumoral and systemic immune biomarkers for locally advanced head-and-neck cancer - detailed protocol of a prospective, observatory multicenter trial (ImmunBioKHT) and first results of the immunophenotyping of the patients' peripheral blood.Front Oncol. 2024 Sep 13;14:1451035. doi: 10.3389/fonc.2024.1451035. eCollection 2024. Front Oncol. 2024. PMID: 39346733 Free PMC article.
-
Inhibition of ATM or ATR in combination with hypo-fractionated radiotherapy leads to a different immunophenotype on transcript and protein level in HNSCC.Front Oncol. 2024 Oct 1;14:1460150. doi: 10.3389/fonc.2024.1460150. eCollection 2024. Front Oncol. 2024. PMID: 39411143 Free PMC article.
-
Clonogenicity-based radioresistance determines the expression of immune suppressive immune checkpoint molecules after hypofractionated irradiation of MDA-MB-231 triple-negative breast cancer cells.Front Oncol. 2023 Apr 20;13:981239. doi: 10.3389/fonc.2023.981239. eCollection 2023. Front Oncol. 2023. PMID: 37152024 Free PMC article.
References
-
- Robert Koch-Institut . Krebs in Deutschland 2015/2016. Robert-Koch-Institut; Berlin, Germany: 2019. Gesellschaft der epidemiologischen Krebsregister in Deutschland e.V. - DOI
-
- Hecht M., Hahn D., Wolber P., Hautmann M.G., Reichert D., Weniger S., Belka C., Bergmann T., Göhler T., Welslau M., et al. Treatment response lowers tumor symptom burden in recurrent and/or metastatic head and neck cancer. BMC Cancer. 2020;20:933. doi: 10.1186/s12885-020-07440-w. - DOI - PMC - PubMed
-
- Sawabe M., Ito H., Oze I., Hosono S., Kawakita D., Tanaka H., Hasegawa Y., Murakami S., Matsuo K. Heterogeneous impact of alcohol consumption according to treatment method on survival in head and neck cancer: A prospective study. Cancer Sci. 2017;108:91–100. doi: 10.1111/cas.13115. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

