Supercritical Antisolvent Fractionation of Antioxidant Compounds from Salvia officinalis
- PMID: 34502255
- PMCID: PMC8431610
- DOI: 10.3390/ijms22179351
Supercritical Antisolvent Fractionation of Antioxidant Compounds from Salvia officinalis
Abstract
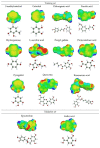
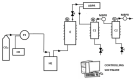
The increasing interest towards greener antioxidants obtained via natural sources and more sustainable processes encourages the development of new theoretical and experimental methods in the field of those compounds. Two advanced separation methods using supercritical CO2 are applied to obtain valuable antioxidants from Salvia officinalis, and a first approximation to a QSAR model relating molecular structure with antioxidant activity is explored in order to be used, in the future, as a guide for the preselection of compounds of interest in these processes. Separation experiments through antisolvent fractionation with supercritical CO2 were designed using a Response Surface Methodology to study the effect of pressure and CO2 flow rate on both mass yields and capability to obtain fractions enriched in three antioxidant compounds: chlorogenic acid, caffeic acid and rosmarinic acid which were tracked using HPLC PDA. Rosmarinic acid was completely retained in the precipitation vessel while chlorogenic and caffeic acids, though distributed between the two separated fractions, had a major presence in the precipitation vessel too. The conditions predicted for an optimal overall yield and enrichment were 148 bar and 10 g/min. Although a training dataset including much more compounds than those now considered can be recommended, descriptors calculated from the σ-profiles provided by COSMO-RS model seem to be adequate for estimating the antioxidant activity of pure compounds through QSAR.
Keywords: QSAR; advanced separation processes; antioxidant activity; caffeic acid; chlorogenic acid; rosmarinic acid; supercritical antisolvent fractionation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Reddy D.N. In: Essential Oils Extracted from Medicinal Plants and Their Applications. Akhtar M.S., Swamy M.K., Sinniah U.R., editors. Springer; Singapore: 2019. pp. 237–283.
-
- Jugreet B.S., Suroowan S., Rengasamy R.R.K., Mahomoodally M.F. Chemistry, Bioactivities, Mode of Action and Industrial Applications of Essential Oils. Trends Food Sci. Technol. 2020;101:89–105. doi: 10.1016/j.tifs.2020.04.025. - DOI
-
- Carpena M., Nunez-Estevez B., Soria-Lopez A., Garcia-Oliveira P., Prieto M.A. Essential Oils and Their Application on Active Packaging Systems: A Review. Resources. 2021;10:7. doi: 10.3390/resources10010007. - DOI
-
- Reverchon E., De Marco I. Supercritical Fluid Extraction and Fractionation of Natural Matter. J. Supercrit. Fluids. 2006;38:146–166. doi: 10.1016/j.supflu.2006.03.020. - DOI
-
- Nagavekar N., Dubey K., Sharma A., Singhal R.S. Innovative Food Processing Technologies. Elsevier; Oxford, UK: 2021. 2.41—Supercritical Extraction of Valued Components from Animals Parts; pp. 597–619.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

