Electrical Detection of Innate Immune Cells
- PMID: 34502775
- PMCID: PMC8433726
- DOI: 10.3390/s21175886
Electrical Detection of Innate Immune Cells
Abstract
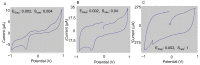
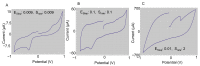
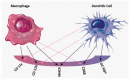
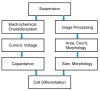
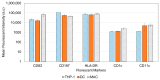
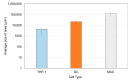
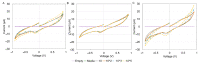
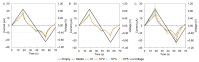
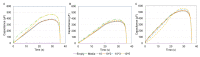
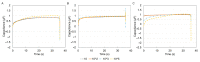
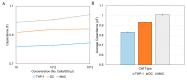
Accurately classifying the innate immune players is essential to comprehensively and quantitatively evaluate the interactions between the innate and the adaptive immune systems. In addition, accurate classification enables the development of models to predict behavior and to improve prospects for therapeutic manipulation of inflammatory diseases and cancer. Rapid development in technologies that provide an accurate definition of the type of cell in action, allows the field of innate immunity to the lead in therapy developments. This article presents a novel immunophenotyping technique using electrical characterization to differentiate between the two most important cell types of the innate immune system: dendritic cells (DCs) and macrophages (MACs). The electrical characterization is based on capacitance measurements, which is a reliable marker for cell surface area and hence cell size. We differentiated THP-1 cells into DCs and MACs in vitro and conducted electrical measurements on the three cell types. The results showed average capacitance readings of 0.83 µF, 0.93 µF, and 1.01 µF for THP-1, DCs, and MACs, respectively. This corresponds to increasing cell size since capacitance is directly proportional to area. The results were verified with image processing. Image processing was used for verification because unlike conventional techniques, especially flow cytometry, it avoids cross referencing and by-passes the limitation of a lack of specificity of markers used to detect the different cell types.
Keywords: dendritic cells; electrical characterization; image processing; immune system; macrophages.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Differential Role of Anti-Viral Sensing Pathway for the Production of Type I Interferon β in Dendritic Cells and Macrophages Against Respiratory Syncytial Virus A2 Strain Infection.Viruses. 2019 Jan 15;11(1):62. doi: 10.3390/v11010062. Viruses. 2019. PMID: 30650519 Free PMC article.
-
[Research advances in the development and function of macrophages and classical dendritic cells during resting and infection].Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2024 Jun;40(6):556-560. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2024. PMID: 38952096 Review. Chinese.
-
A phased strategy to differentiate human CD14+monocytes into classically and alternatively activated macrophages and dendritic cells.Biotechniques. 2016 Jul 1;61(1):33-41. doi: 10.2144/000114435. eCollection 2016. Biotechniques. 2016. PMID: 27401672 Free PMC article.
-
The impact of eicosanoids on the crosstalk between innate and adaptive immunity: the key roles of dendritic cells.Tissue Antigens. 2005 Jun;65(6):507-14. doi: 10.1111/j.1399-0039.2005.00394.x. Tissue Antigens. 2005. PMID: 15896197 Review.
-
Unexpected mechanism of colitis amelioration by artesunate, a natural product from Artemisia annua L.Inflammopharmacology. 2020 Aug;28(4):851-868. doi: 10.1007/s10787-019-00678-2. Epub 2019 Dec 21. Inflammopharmacology. 2020. PMID: 31865495
Cited by
-
Special Issue "Computer Aided Diagnosis Sensors".Sensors (Basel). 2022 Oct 21;22(20):8052. doi: 10.3390/s22208052. Sensors (Basel). 2022. PMID: 36298403 Free PMC article.
References
-
- Macrophage—An Overview. ScienceDirect Topics. [(accessed on 1 July 2020)]; Available online: https://www.sciencedirect.com/topics/materials-science/macrophage.
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

