Pharmacologic therapy for engraftment arrhythmia induced by transplantation of human cardiomyocytes
- PMID: 34506727
- PMCID: PMC8514851
- DOI: 10.1016/j.stemcr.2021.08.005
Pharmacologic therapy for engraftment arrhythmia induced by transplantation of human cardiomyocytes
Abstract
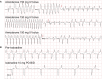
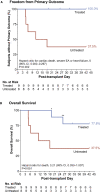
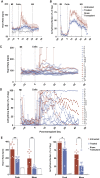
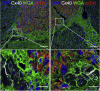
Heart failure remains a significant cause of morbidity and mortality following myocardial infarction. Cardiac remuscularization with transplantation of human pluripotent stem cell-derived cardiomyocytes is a promising preclinical therapy to restore function. Recent large animal data, however, have revealed a significant risk of engraftment arrhythmia (EA). Although transient, the risk posed by EA presents a barrier to clinical translation. We hypothesized that clinically approved antiarrhythmic drugs can prevent EA-related mortality as well as suppress tachycardia and arrhythmia burden. This study uses a porcine model to provide proof-of-concept evidence that a combination of amiodarone and ivabradine can effectively suppress EA. None of the nine treated subjects experienced the primary endpoint of cardiac death, unstable EA, or heart failure compared with five out of eight (62.5%) in the control cohort (hazard ratio = 0.00; 95% confidence interval: 0-0.297; p = 0.002). Pharmacologic treatment of EA may be a viable strategy to improve safety and allow further clinical development of cardiac remuscularization therapy.
Keywords: antiarrhythmic drugs; cardiac cell therapy; cardiac regeneration; cardiac remuscularization; electrophysiology; embryonic stem cells; engraftment arrhythmia; heart failure; myocardial infarction; sudden cardiac death.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Figures


References
-
- Bedada F.B., Chan S.S., Metzger S.K., Zhang L., Zhang J., Garry D.J., Kamp T.J., Kyba M., Metzger J.M. Acquisition of a quantitative, stoichiometrically conserved ratiometric marker of maturation status in stem cell-derived cardiac myocytes. Stem Cell Reports. 2014;3:594–605. doi: 10.1016/j.stemcr.2014.07.012. - DOI - PMC - PubMed
-
- Caspi O., Huber I., Kehat I., Habib M., Arbel G., Gepstein A., Yankelson L., Aronson D., Beyar R., Gepstein L. Transplantation of human embryonic stem cell-derived cardiomyocytes improves myocardial performance in infarcted rat hearts. J. Am. Coll. Cardiol. 2007;50:1884–1893. doi: 10.1016/j.jacc.2007.07.054. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

