Detection of Neoplasms by Metagenomic Next-Generation Sequencing of Cerebrospinal Fluid
- PMID: 34515766
- PMCID: PMC8438621
- DOI: 10.1001/jamaneurol.2021.3088
Detection of Neoplasms by Metagenomic Next-Generation Sequencing of Cerebrospinal Fluid
Abstract
Importance: Cerebrospinal fluid (CSF) cytologic testing and flow cytometry are insensitive for diagnosing neoplasms of the central nervous system (CNS). Such clinical phenotypes can mimic infectious and autoimmune causes of meningoencephalitis.
Objective: To ascertain whether CSF metagenomic next-generation sequencing (mNGS) can identify aneuploidy, a hallmark of malignant neoplasms, in difficult-to-diagnose cases of CNS malignant neoplasm.
Design, setting, and participants: Two case-control studies were performed at the University of California, San Francisco (UCSF). The first study used CSF specimens collected at the UCSF Clinical Laboratories between July 1, 2017, and December 31, 2019, and evaluated test performance in specimens from patients with a CNS malignant neoplasm (positive controls) or without (negative controls). The results were compared with those from CSF cytologic testing and/or flow cytometry. The second study evaluated patients who were enrolled in an ongoing prospective study between April 1, 2014, and July 31, 2019, with presentations that were suggestive of neuroinflammatory disease but who were ultimately diagnosed with a CNS malignant neoplasm. Cases of individuals whose tumors could have been detected earlier without additional invasive testing are discussed.
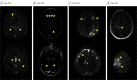
Main outcomes and measures: The primary outcome measures were the sensitivity and specificity of aneuploidy detection by CSF mNGS. Secondary subset analyses included a comparison of CSF and tumor tissue chromosomal abnormalities and the identification of neuroimaging characteristics that were associated with test performance.
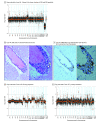
Results: Across both studies, 130 participants were included (median [interquartile range] age, 57.5 [43.3-68.0] years; 72 men [55.4%]). The test performance study used 125 residual laboratory CSF specimens from 47 patients with a CNS malignant neoplasm and 56 patients with other neurological diseases. The neuroinflammatory disease study enrolled 12 patients and 17 matched control participants. The sensitivity of the CSF mNGS assay was 75% (95% CI, 63%-85%), and the specificity was 100% (95% CI, 96%-100%). Aneuploidy was detected in 64% (95% CI, 41%-83%) of the patients in the test performance study with nondiagnostic cytologic testing and/or flow cytometry, and in 55% (95% CI, 23%-83%) of patients in the neuroinflammatory disease study who were ultimately diagnosed with a CNS malignant neoplasm. Of the patients in whom aneuploidy was detected, 38 (90.5%) had multiple copy number variations with tumor fractions ranging from 31% to 49%.
Conclusions and relevance: This case-control study showed that CSF mNGS, which has low specimen volume requirements, does not require the preservation of cell integrity, and was orginally developed to diagnose neurologic infections, can also detect genetic evidence of a CNS malignant neoplasm in patients in whom CSF cytologic testing and/or flow cytometry yielded negative results with a low risk of false-positive results.
Conflict of interest statement
Figures


References
-
- Granerod J, Ambrose HE, Davies NW, et al. ; UK Health Protection Agency (HPA) Aetiology of Encephalitis Study Group . Causes of encephalitis and differences in their clinical presentations in England: a multicentre, population-based prospective study. Lancet Infect Dis. 2010;10(12):835-844. doi: 10.1016/S1473-3099(10)70222-X - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

