Proportion of Women and Reporting of Outcomes by Sex in Clinical Trials for Alzheimer Disease: A Systematic Review and Meta-analysis
- PMID: 34515784
- PMCID: PMC12243614
- DOI: 10.1001/jamanetworkopen.2021.24124
Proportion of Women and Reporting of Outcomes by Sex in Clinical Trials for Alzheimer Disease: A Systematic Review and Meta-analysis
Abstract
Importance: Women represent two-thirds of patients with Alzheimer disease (AD), and sex differences might affect results of randomized clinical trials (RCTs). However, little information exists on differences in sex as reported in RCTs for AD.
Objective: To assess the ratio of females to males and the reporting of sex-stratified data in large pharmaceutical RCTs for AD.
Data sources: A search for pharmaceutical RCTs for AD was conducted on September 4, 2019, using ClinicalTrials.gov with the key word Alzheimer disease, and articles related to those trials were identified using the PubMed, Scopus, and Google Scholar databases. Searches were conducted between September 4 and October 31, 2019, and between April 15 and May 31, 2020.
Study selection: Controlled RCTs that had more than 100 participants and tested the efficacy of drugs or herbal extracts were included. Of 1047 RCTs identified, 409 were published and therefore screened. A total of 77 articles were included in the final analysis, including 56 primary articles on AD, 13 secondary articles on AD, and 8 articles on mild cognitive impairment.
Data extraction and synthesis: The location and date of publication; number, sex, and age of patients enrolled; disease severity; experimental or approved status of the drug; and whether the study included a sex-stratified analysis in the protocol, methods, or results were extracted by 1 reviewer for each article, and the meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guideline. Data were analyzed using a mixed-effects model.
Main outcomes and measures: The mean proportion of women enrolled in the trials and the associations between prespecified variables were analyzed. The proportion of articles that included sex-stratified results and the temporal trends in the reporting of these results were also studied.
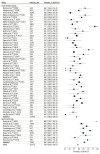
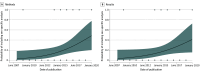
Results: In this review of 56 RCTs for AD involving 39 575 participants, 23 348 women (59.0%) were included. The mean (SD) proportion of women in RCTs of approved drugs was 67.3% (6.9%), and in RCTs of experimental drugs was 57.9% (5.9%). The proportion of women in RCTs of experimental drugs was significantly lower than the proportion of women in the general population with AD in the US (62.1%; difference, -4.56% [95% CI, -6.29% to -2.87%]; P < .001) and Europe (68.2%; difference, -10.67% [95% CI, -12.39% to -8.97%]; P < .001). Trials of approved drugs had a higher probability of including women than trials of experimental drugs (odds ratio [OR], 1.26; 95% CI, 1.05-1.52; P = .02). Both the severity of AD at baseline and the trial location were associated with the probability of women being enrolled in trials (severity: OR, 0.98; 95% CI, 0.97-1.00; P = .02; location in Europe: OR, 1.26; 95% CI, 1.05-1.52; P = .01; location in North America: OR, 0.81; 95% CI, 0.71-0.93; P = .002). Only 7 articles (12.5%) reported sex-stratified results, with an increasing temporal trend (R, 0.30; 95% CI, 0.05-0.59; P = .03).
Conclusions and relevance: In this systematic review and meta-analysis, the proportion of women in RCTs for AD, although higher than the proportion of men, was significantly lower than that in the general population. Only a small proportion of trials reported sex-stratified results. These findings support strategies to improve diversity in enrollment and data reporting in RCTs for AD.
Conflict of interest statement
Figures
Comment in
-
Treatment for Alzheimer Disease-Sex and Gender Effects Need to Be Explicitly Analyzed and Reported in Clinical Trials.JAMA Netw Open. 2021 Sep 1;4(9):e2124386. doi: 10.1001/jamanetworkopen.2021.24386. JAMA Netw Open. 2021. PMID: 34515788 No abstract available.
References
-
- Prince M, Wimo A, Guerchet M, Ali GC, Wu YT, Prina M. World Alzheimer Report 2015: the global impact of dementia—an analysis of prevalence, incidence, cost and trends. Alzheimer’s Disease International. 2015. Accessed June 15, 2020. https://www.alzint.org/resource/world-alzheimer-report-2015/
-
- Alzheimer Europe. Dementia in Europe Yearbook 2019: Estimating the Prevalence of Dementia in Europe. Alzheimer Europe; 2019.
-
- Alzheimer’s Association. 2020 Alzheimer’s disease facts and figures. Alzheimers Dement. 2020;16(3):391–460. doi:10.1002/alz.12068 doi: 10.1002/alz.12068 - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

