Fat causes necrosis and inflammation in parenchymal cells in human steatotic liver
- PMID: 34524512
- PMCID: PMC8755686
- DOI: 10.1007/s00418-021-02030-8
Fat causes necrosis and inflammation in parenchymal cells in human steatotic liver
Abstract
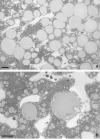
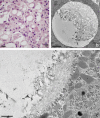
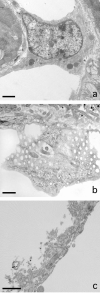
Adapted fixation methods for electron microscopy allowed us to study liver cell fine structure in 217 biopsies of intact human livers over the course of 10 years. The following novel observations and concepts arose: single fat droplets in parenchymal cells can grow to a volume four times larger than the original cell, thereby extremely marginalizing the cytoplasm with all organelles. Necrosis of single parenchymal cells, still containing one huge fat droplet, suggests death by fat in a process of single-cell steatonecrosis. In a later stage of single-cell steatonecrosis, neutrophils and erythrocytes surround the single fat droplet, forming an inflammatory fat follicle indicating the apparent onset of inflammation. Also, fat droplets frequently incorporate masses of filamentous fragments and other material, most probably representing Mallory substance. No other structure or material was found that could possibly represent Mallory bodies. We regularly observe the extrusion of huge fat droplets, traversing the peripheral cytoplasm of parenchymal cells, the Disse space and the endothelium. These fat droplets fill the sinusoid as a sinusoidal lipid embolus. In conclusion, adapted methods of fixation applied to human liver tissue revealed that single, huge fat droplets cause necrosis and inflammation in single parenchymal cells. Fat droplets also collect Mallory substance and give rise to sinusoidal fat emboli. Therefore, degreasing of the liver seems to be an essential therapeutic first step in the self-repairing of non-alcoholic fatty liver disease. This might directly reduce single-cell steatotic necrosis and inflammation as elements in non-alcoholic steatohepatitis progression.
Keywords: Fat; Human liver; Inflammation; Necrosis; Parenchymal cell; Steatosis.
© 2021. The Author(s).
Figures



Similar articles
-
Cooperation of liver cells in health and disease.Adv Anat Embryol Cell Biol. 2001;161:III-XIII, 1-151. doi: 10.1007/978-3-642-56553-3. Adv Anat Embryol Cell Biol. 2001. PMID: 11729749 Review.
-
Pediatric non-alcoholic steatohepatitis: the first report on the ultrastructure of hepatocyte mitochondria.World J Gastroenterol. 2014 Apr 21;20(15):4335-40. doi: 10.3748/wjg.v20.i15.4335. World J Gastroenterol. 2014. PMID: 24764670 Free PMC article.
-
Adipogenic changes of hepatocytes in a high-fat diet-induced fatty liver mice model and non-alcoholic fatty liver disease patients.Endocrine. 2015 Apr;48(3):834-47. doi: 10.1007/s12020-014-0384-x. Epub 2014 Aug 20. Endocrine. 2015. PMID: 25138963
-
Non-Alcoholic Fatty Liver Disease.Adv Exp Med Biol. 2017;960:443-467. doi: 10.1007/978-3-319-48382-5_19. Adv Exp Med Biol. 2017. PMID: 28585211 Review.
-
Pathogenesis of NASH: How Metabolic Complications of Overnutrition Favour Lipotoxicity and Pro-Inflammatory Fatty Liver Disease.Adv Exp Med Biol. 2018;1061:19-44. doi: 10.1007/978-981-10-8684-7_3. Adv Exp Med Biol. 2018. PMID: 29956204 Review.
Cited by
-
Self-Organization of Sinusoidal Vessels in Pluripotent Stem Cell-derived Human Liver Bud Organoids.bioRxiv [Preprint]. 2024 Jul 4:2024.07.02.601804. doi: 10.1101/2024.07.02.601804. bioRxiv. 2024. Update in: Nat Biomed Eng. 2025 Jun 25. doi: 10.1038/s41551-025-01416-6. PMID: 39005378 Free PMC article. Updated. Preprint.
-
In focus in HCB.Histochem Cell Biol. 2022 Jan;157(1):1-5. doi: 10.1007/s00418-021-02064-y. Histochem Cell Biol. 2022. PMID: 34981200 No abstract available.
-
Comparative analysis of high-fat diets: Effects of mutton, beef, and vegetable fats on body weight, biochemical profiles, and liver histology in mice.Heliyon. 2024 Oct 12;10(20):e39349. doi: 10.1016/j.heliyon.2024.e39349. eCollection 2024 Oct 30. Heliyon. 2024. PMID: 39640651 Free PMC article.
-
Mechanical rheological model on the assessment of elasticity and viscosity in tissue inflammation: A systematic review.PLoS One. 2024 Jul 15;19(7):e0307113. doi: 10.1371/journal.pone.0307113. eCollection 2024. PLoS One. 2024. PMID: 39008477 Free PMC article.
-
Portal hypertension in patients with nonalcoholic fatty liver disease: Current knowledge and challenges.World J Gastroenterol. 2024 Jan 28;30(4):290-307. doi: 10.3748/wjg.v30.i4.290. World J Gastroenterol. 2024. PMID: 38313235 Free PMC article. Review.
References
-
- Arias IM. The biology of hepatic endothelial cell fenestrae. Prog Liver Dis. 1990;9:11–26. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

