Unravelling the antimicrobial activity of peptide hydrogel systems: current and future perspectives
- PMID: 34525154
- PMCID: PMC8442837
- DOI: 10.1039/d1sm00839k
Unravelling the antimicrobial activity of peptide hydrogel systems: current and future perspectives
Abstract
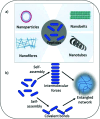
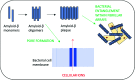
The use of hydrogels has garnered significant interest as biomaterial and drug delivery platforms for anti-infective applications. For decades antimicrobial peptides have been heralded as a much needed new class of antimicrobial drugs. Self-assembling peptide hydrogels with inherent antimicrobial ability have recently come to the fore. However, their fundamental antimicrobial properties, selectivity and mechanism of action are relatively undefined. This review attempts to establish a link between antimicrobial efficacy; the self-assembly process; peptide-membrane interactions and mechanical properties by studying several reported peptide systems: β-hairpin/β-loop peptides; multidomain peptides; amphiphilic surfactant-like peptides and ultrashort/low molecular weight peptides. We also explore their role in the formation of amyloid plaques and the potential for an infection etiology in diseases such as Alzheimer's. We look briefly at innovative methods of gel characterization. These may provide useful tools for future studies within this increasingly important field.
Conflict of interest statement
There are no conflicts to declare.
Figures







References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

