Cerebellar complex spikes multiplex complementary behavioral information
- PMID: 34529650
- PMCID: PMC8478165
- DOI: 10.1371/journal.pbio.3001400
Cerebellar complex spikes multiplex complementary behavioral information
Abstract
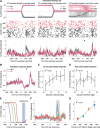
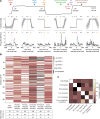
Purkinje cell (PC) discharge, the only output of cerebellar cortex, involves 2 types of action potentials, high-frequency simple spikes (SSs) and low-frequency complex spikes (CSs). While there is consensus that SSs convey information needed to optimize movement kinematics, the function of CSs, determined by the PC's climbing fiber input, remains controversial. While initially thought to be specialized in reporting information on motor error for the subsequent amendment of behavior, CSs seem to contribute to other aspects of motor behavior as well. When faced with the bewildering diversity of findings and views unraveled by highly specific tasks, one may wonder if there is just one true function with all the other attributions wrong? Or is the diversity of findings a reflection of distinct pools of PCs, each processing specific streams of information conveyed by climbing fibers? With these questions in mind, we recorded CSs from the monkey oculomotor vermis deploying a repetitive saccade task that entailed sizable motor errors as well as small amplitude saccades, correcting them. We demonstrate that, in addition to carrying error-related information, CSs carry information on the metrics of both primary and small corrective saccades in a time-specific manner, with changes in CS firing probability coupled with changes in CS duration. Furthermore, we also found CS activity that seemed to predict the upcoming events. Hence PCs receive a multiplexed climbing fiber input that merges complementary streams of information on the behavior, separable by the recipient PC because they are staggered in time.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures






References
-
- Albus JS. A theory of cerebellar function. Math Biosci. 1971;10(1–2):25–61.
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources

