Evidence-based clinical practice guidelines for nonalcoholic fatty liver disease/nonalcoholic steatohepatitis 2020
- PMID: 34533632
- PMCID: PMC8531062
- DOI: 10.1007/s00535-021-01796-x
Evidence-based clinical practice guidelines for nonalcoholic fatty liver disease/nonalcoholic steatohepatitis 2020
Abstract
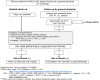
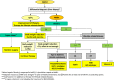
Nonalcoholic fatty liver disease (NAFLD) has become a serious public health issue not only in Western countries but also in Japan. Within the wide spectrum of NAFLD, nonalcoholic steatohepatitis (NASH) is a progressive form of disease that often develops into liver cirrhosis and increases the risk of hepatocellular carcinoma (HCC). While a definite diagnosis of NASH requires liver biopsy to confirm the presence of hepatocyte ballooning, hepatic fibrosis is the most important prognostic factor in NAFLD. With so many NAFLD patients, it is essential to have an effective screening method for NAFLD with hepatic fibrosis. As HCC with non-viral liver disease has increased markedly in Japan, effective screening and surveillance of HCC are also urgently needed. The most common death etiology in NAFLD patients is cardiovascular disease (CVD) event. Gastroenterologists must, therefore, pay close attention to CVD when examining NAFLD patients. In the updated guidelines, we propose screening and follow-up methods for hepatic fibrosis, HCC, and CVD in NAFLD patients. Several drug trials are ongoing for NAFLD/NASH therapy, however, there is currently no specific drug therapy for NAFLD/NASH. In addition to vitamin E and thiazolidinedione derivatives, recent trials have focused on sodium glucose co-transporter 2 (SGLT2) inhibitors and glucagon-like peptide-1 (GLP-1) analogues, and effective therapies are expected to be developed. These practical guidelines for NAFLD/NASH were established by the Japanese Society of Gastroenterology in conjunction with the Japan Society of Hepatology. Clinical evidence reported internationally between 1983 and October 2018 was collected, and each clinical and background question was evaluated using the Grades of Recommendation Assessment, Development and Evaluation (GRADE) system. This English summary provides the core essentials of these clinical practice guidelines, which include the definition and concept, screening systems for hepatic fibrosis, HCC and CVD, and current therapies for NAFLD/NASH in Japan.
Keywords: Cardiovascular disease; Fibrosis; HCC; NAFLD/NASH guidelines; Treatment.
© 2021. The Author(s).
Conflict of interest statement
Any financial relationship with enterprises, businesses, or academic institutions in the subject matter or materials discussed in the manuscript are listed as follows: (1) those from which the authors, the spouse, partner or immediate relatives of authors, have received individually any income, honoraria, or any other types of remuneration—MSD, Otsuka Pharmaceutical, Mitsubishi Tanabe Pharma, Bristol-Myers Squibb, Lundbeck Japan, AbbVie, Gilead Sciences, Sumitomo Dainippon Pharma, Novo Nordisk, EA Pharma, Astellas Pharma, Mylan, Mochida Pharmaceutical, Kowa Company, Tsumura & CO., and (2) those from which the academic institutions of the authors received support (commercial/academic cooperation)—EA Pharma, Astellas Pharma, Eisai, MSD, Otsuka Pharmaceutical, Suntory Global Innovation Center, Shionogi & Co., Daiichi Sankyo, Sumitomo Dainippon Pharma, Taiho Pharmaceutical, Takeda Pharmaceutical, Mitsubishi Tanabe Pharma, Chugai Pharmaceutical, Bayer Yakuhin, Bristol-Myers Squibb, Mochida Pharmaceutical, Yakult Honsha, AbbVie, Gilead Sciences, Gadelius Medical, Novartis Pharma, Eli Lilly Japan, Mylan N.V., Century Medical, Sogo Rinsho Medefi, Nissan Chemical, Fujirebio. Kowa, Kyokuto Pharmaceutical Industrial. Biofermin Pharmaceutical, and (3) those from which the authors have received individually endowed chair—NichiNichi Pharmaceutical.
Figures


References
-
- EASL-EASD-EASO Clinical Practice Guidelines for the management of non- alcoholic fatty liver disease. J Hepatol. 2016;64:1388–1402. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

