Bacterial and Host Determinants of Group B Streptococcal Vaginal Colonization and Ascending Infection in Pregnancy
- PMID: 34540718
- PMCID: PMC8446444
- DOI: 10.3389/fcimb.2021.720789
Bacterial and Host Determinants of Group B Streptococcal Vaginal Colonization and Ascending Infection in Pregnancy
Abstract
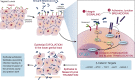
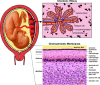
Group B streptococcus (GBS) is a gram-positive bacteria that asymptomatically colonizes the vaginal tract. However, during pregnancy maternal GBS colonization greatly predisposes the mother and baby to a wide range of adverse outcomes, including preterm birth (PTB), stillbirth, and neonatal infection. Although many mechanisms involved in GBS pathogenesis are partially elucidated, there is currently no approved GBS vaccine. The development of a safe and effective vaccine that can be administered during or prior to pregnancy remains a principal objective in the field, because current antibiotic-based therapeutic strategies do not eliminate all cases of invasive GBS infections. Herein, we review our understanding of GBS disease pathogenesis at the maternal-fetal interface with a focus on the bacterial virulence factors and host defenses that modulate the outcome of infection. We follow GBS along its path from an asymptomatic colonizer of the vagina to an invasive pathogen at the maternal-fetal interface, noting factors critical for vaginal colonization, ascending infection, and vertical transmission to the fetus. Finally, at each stage of infection we emphasize important host-pathogen interactions, which, if targeted therapeutically, may help to reduce the global burden of GBS.
Keywords: bacteria; colonization; fetus; group B streptococcus; placenta; pregnancy; preterm birth; vagina.
Copyright © 2021 Brokaw, Furuta, Dacanay, Rajagopal and Adams Waldorf.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Absalon J., Segall N., Block S. L., Center K. J., Scully I. L., Giardina P. C., et al. . (2021). Safety and Immunogenicity of a Novel Hexavalent Group B Streptococcus Conjugate Vaccine in Healthy, Non-Pregnant Adults: A Phase 1/2, Randomised, Placebo-Controlled, Observer-Blinded, Dose-Escalation Trial. Lancet Infect. Dis. 21, 263–274. 10.1016/S1473-3099(20)30478-3 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

