Temporo-cerebellar connectivity underlies timing constraints in audition
- PMID: 34542407
- PMCID: PMC8480974
- DOI: 10.7554/eLife.67303
Temporo-cerebellar connectivity underlies timing constraints in audition
Abstract
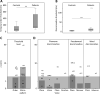
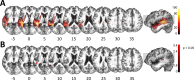
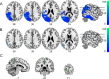
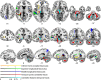
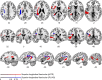
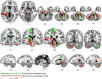
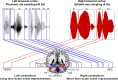
The flexible and efficient adaptation to dynamic, rapid changes in the auditory environment likely involves generating and updating of internal models. Such models arguably exploit connections between the neocortex and the cerebellum, supporting proactive adaptation. Here, we tested whether temporo-cerebellar disconnection is associated with the processing of sound at short timescales. First, we identify lesion-specific deficits for the encoding of short timescale spectro-temporal non-speech and speech properties in patients with left posterior temporal cortex stroke. Second, using lesion-guided probabilistic tractography in healthy participants, we revealed bidirectional temporo-cerebellar connectivity with cerebellar dentate nuclei and crura I/II. These findings support the view that the encoding and modeling of rapidly modulated auditory spectro-temporal properties can rely on a temporo-cerebellar interface. We discuss these findings in view of the conjecture that proactive adaptation to a dynamic environment via internal models is a generalizable principle.
Keywords: audition; human; internal models; lateralization; lesion mapping; neuroscience; temporo-cerebellar connectivity; tractography.
© 2021, Stockert et al.
Conflict of interest statement
AS, MS, DP, AA, SK No competing interests declared
Figures



References
-
- Anstis SM, Atkinson J, Blakemore C, Braddick O, Brandt T, Campbell FW, Coren S, Dichgans J, Dodwell PC, Eimas PD, Foley JM, Fox R, Ganz L, Garrett M, Gibson EJ, Girgus JS, Haith MM, Hatwell Y, Hilgard ER, Ingle D, Johansson G, Julesz B, Konishi M, Lackner JR, Levinson E, Liberman AM, Maffei L, Oyama T, Pantle A, Pöppel E, Sekuler R, Stromeyer CF, Studdert-Kennedy M, Teuber H-L, Yin RK, Held R, Leibowitz HW, Teuber H-L. In: Perception. Held R, Leibowitz HW, Teuber HL, editors. Berlin, Heidelberg: Springer Berlin Heidelberg; 1978. Perception; pp. 713–729. - DOI
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources

