Risk Factors Associated with Durable Progression-Free Survival in Patients with Relapsed or Refractory Multiple Myeloma Treated with Anti-BCMA CAR T-cell Therapy
- PMID: 34548316
- PMCID: PMC9401500
- DOI: 10.1158/1078-0432.CCR-21-2031
Risk Factors Associated with Durable Progression-Free Survival in Patients with Relapsed or Refractory Multiple Myeloma Treated with Anti-BCMA CAR T-cell Therapy
Abstract
Purpose: B-cell maturation antigen (BCMA) chimeric antigen receptor (CAR) T-cell therapy results in high remission rates in patients with relapsed/refractory (R/R) multiple myeloma. However, the factors associated with prognosis following CAR T-cell therapy are unknown.
Patients and methods: Between July 1, 2018 and July 31, 2020, 61 patients with R/R multiple myeloma received anti-BCMA CAR T-cell therapy (Chictr.org number, ChiCTR1800017404). Step-wise multivariate Cox regression and competing risk analyses were conducted to identify poor prognosis-associated risk factors.
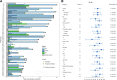
Results: Sixty patients (98.4%) experienced cytokine release syndrome (CRS), including 33, 23, and 4 cases of CRS grades 1 to 2, 3, and 4, respectively. The objective response rate (ORR) was 98.3%, and the complete remission (CR) rate was 70.3%. With a median follow-up period of 21.1 months, the 1-year overall survival (OS) and progression-free survival (PFS) rates were 78.0% and 50.2%, respectively. The median PFS was 12.7 months. Cox modeling revealed that poor PFS was associated with extramedullary disease [HR = 2.59, 95% confidence interval (95% CI) = 1.29-5.21, P = 0.008], light chain multiple myeloma (HR = 2.53, 95% CI = 1.03-5.97, P = 0.035), high-risk cytogenetics (HR = 2.80, 95% CI = 1.27-6.14, P = 0.01), and prior treatment with more than 3 therapeutic lines (HR = 3.14, 95% CI = 1.34-7.34, P = 0.008). Among the 41 CR cases, competing risk analyses demonstrated higher relapse predispositions in those with extramedullary disease (HR = 4.51, 95% CI = 1.86-10.9, P = 0.001), light chain multiple myeloma (HR = 4.89, 95% CI = 1.52 - 15.7, P = 0.008), or high-risk cytogenetics (HR = 5.09, 95% CI = 1.63-15.9, P = 0.005).
Conclusions: Anti-BCMA CAR T-cell therapy is safe and effective for R/R multiple myeloma. For patients with high-risk factors, improvements to extend remission and more specific individualized therapies are needed.
©2021 The Authors; Published by the American Association for Cancer Research.
Figures



Comment on
-
Selected Articles from This Issue.Clin Cancer Res. 2021 Dec 1;27(23):6279. doi: 10.1158/1078-0432.CCR-27-23-HI. Clin Cancer Res. 2021. PMID: 34853073 No abstract available.
References
-
- Röllig C, Knop S, Bornhäuser M. Multiple myeloma. Lancet 2015;385:2197–208. - PubMed
-
- Kumar SK, Rajkumar V, Kyle RA, van Duin M, Sonneveld P, Mateos MV, et al. . Multiple myeloma. Nat Rev Dis Primers 2017;3:17046. - PubMed
-
- Kumar S. Treatment of newly diagnosed multiple myeloma in transplant-eligible patients. Curr Hematol Malig Rep 2011;6:104–12. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

