Role of glypican-1 in regulating multiple cellular signaling pathways
- PMID: 34550795
- PMCID: PMC8616591
- DOI: 10.1152/ajpcell.00290.2021
Role of glypican-1 in regulating multiple cellular signaling pathways
Abstract
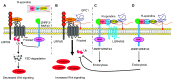
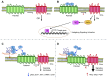
Glypican-1 (GPC1) is one of the six glypican family members in humans. It is composed of a core protein with three heparan sulfate chains and attached to the cell membrane by a glycosyl-phosphatidylinositol anchor. GPC1 modulates various signaling pathways including fibroblast growth factors (FGF), vascular endothelial growth factor-A (VEGF-A), transforming growth factor-β (TGF-β), Wnt, Hedgehog (Hh), and bone morphogenic protein (BMP) through specific interactions with pathway ligands and receptors. The impact of these interactions on signaling pathways, activating or inhibitory, is dependent upon specific GPC1 domain interaction with pathway components, as well as cell surface context. In this review, we summarize the current understanding of the structure of GPC1, as well as its role in regulating multiple signaling pathways. We focus on the functions of GPC1 in cancer cells and how new insights into these signaling processes can inform its translational potential as a therapeutic target in cancer.
Keywords: cancer; glypican-1; signaling pathways; therapeutic target.
Conflict of interest statement
The authors are inventors on international patent applications no. PCT/US2020/013739, “High affinity monoclonal antibodies targeting glypican-1 and methods of use thereof.” The authors declare no other conflicts of interest. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government. The antibodies targeting glypican-1 (GPC1) are available for licensing, in a wide range of fields of use, from the National Cancer Institute, NIH. If you are interested in obtaining a license, please contact the principal investigator Dr. Mitchell Ho at
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

